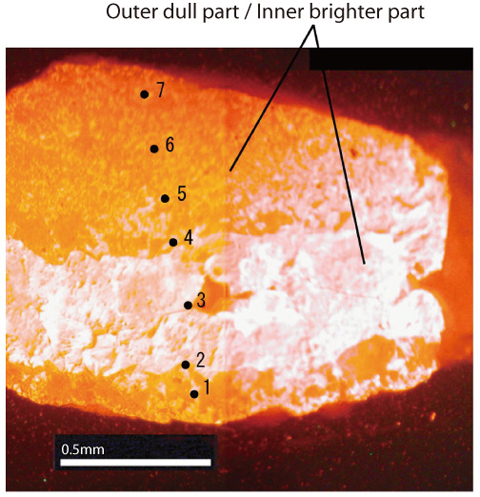
Fig.2-14 Cathode luminescence image of sampled calcite
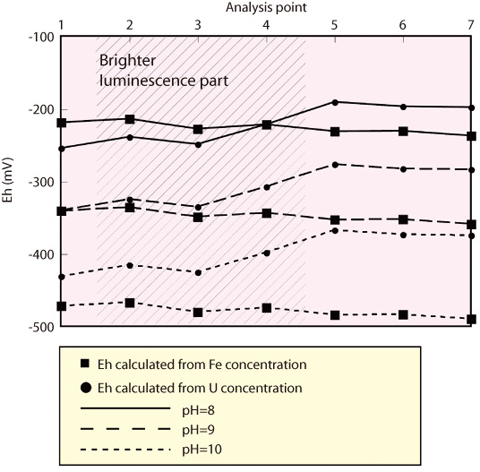
Fig.2-15 Calculation result
With the research and development for the long-term disposal of radioactive wastes, it is necessary to understand the nature of mass transport in natural environments. This will depend on the groundwater chemistry. Thus, evolution processes and long-term stability of groundwater chemistry should be understood to predict the change of hydrochemical conditions in the future.
In this study, to develop a methodology for assessing the paleo-hydrochemical evolution process of groundwater, especially with respect to understanding variations in redox conditions, calcite was studied. Chemical data from calcite have been found to be particularly useful for understanding chemical evolution processes of groundwater, which may affect the precipitation of calcite and thus be recorded. Such change of chemical environment will be recorded as a zonal structure in calcite grain (Fig.2-14). To calculate evolution of potential, firstly, concentration of metal element in past groundwater was calculated based on their concentration in calcite using partition coefficient. Next, redox potential was calculated based on the redox equilibrium of the metal element. In this way, we can guess the redox potential when calcite was precipitated.
Observation and quantitative analysis were carried out using 6 fracture filling calcite samples taken from basement granite. Calculated paleo-redox potential based on the concentrations of U and Fe are similar to present redox conditions (Eh: approx. -270mV to -400mV) in the case of pH=8. Based on this result, it is suggested that the redox conditions of the calcite in the evolving groundwater have varied with a range of several tens mV (Fig.2-15). Thus, the present study suggests that redox conditions in this area may have changed little. Therefore, the redox condition of groundwater likely will remain in reduction condition during calcite precipitation.
There has been no research which appraises the change of redox potential quantitatively, as in this research. This new understanding of natural phenomenon can contribute to a more realistic setting of conditions for mass transfer analysis, an so should be important knowledge in safety assessment of disposal of radioactive wastes.