
Fig.14-26 DOM oxidation reaction vessel

Fig.14-27 CO2 purification glass line
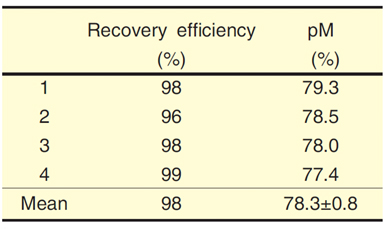
Table14-2 Experimental precision testing using surface seawater
An Accelerator Mass Spectrometry (AMS) can measure extremely small amounts of long half-life radionuclides with high sensitivity and high precision. An AMS system with the ability to measure radiocarbon (14C) and radioiodine (129I) has been installed in the Mutsu Office of the Aomori Research and Development Center. The characteristics of AMS allow it to be used not only in fields closely related to nuclear power, such as for environmental radiation measurements, but also in various fields such as earth sciences and archaeology.
In recent years, global warming has become a serious problem and a more accurate understanding of the earth's carbon cycle is being sought. Because 14C (half-life 5,730 years) can serve as a time-scale index for this carbon cycle, it can be said that 14C measurements using AMS and building a global scale 14C database is the key to resolving the carbon cycle mechanism. The organic matter fixed by photosynthesis from atmospheric carbon dioxide (CO2) plays an important role in the carbon cycle. However, 14C research on the portion of organic matter that is in the form of dissolved organic matter (DOM) in seawater has been extremely small compared to 14C research on terrestrial organic matter or particulate organic matter in seawater. This is because the experimental techniques required to measure 14C in DOM in seawater are difficult, as great effort is required for shipboard filtering of large amounts of seawater (4L), and from samples that contain a salinity more than 10,000 times the concentration of DOM, it is difficult to extract the carbon only for the 14C measurements. Particularly for the ocean around Japan, there are no examples of research. DOM in seawater accounts for about 20% of the organic matter on the earth's surface and more than 90% of the organic matter in the ocean. In terms of carbon, this amount rivals that of atmospheric CO2. Therefore, it is extremely important when considering the carbon cycle on the surface of the earth. Therefore, there is a great interest from this perspective in the development of AMS measurement methods for 14C in seawater DOM.
To resolve these issues, we have established a method to decompose DOM using ultraviolet (UV) irradiation that can be used with high salinity samples and have combined that with AMS. The two-step decomposition method involves first irradiating DOM samples with UV to produce CO2 (Fig.14-26) that is then collected and purified (Fig.14-27). Our study of the UV irradiation time enabled the recovery efficiency to exceed 96%. Improvements to the reaction vessel and glass lines allowed exclusion of all CO2 other than that from the sample, and we were successful in reducing contamination to less than 1% of the sample (world's highest standard). When standard samples of organic matter were decomposed using this method, the results for 14C measurement using AMS had a precision of 0.2% (n = 10). These results mean that it is possible to measure the time scale for DOM with a small measurement error of a decade or so. Experiments with actual seawater showed that an adequate recovery efficiency (98%) was obtained (Table 14-2). Furthermore, for DOM in seawater (oldest samples being about 6,000 years old) that has a low 14C compared to other environmental samples, it was verified that a highly replicable result (0.8%: a period of about 80 years) was obtained.
As DOM affects (and is affected by) all biological activities on the earth, there is interest in this method from researchers in many fields. Measurement of 14C in DOM using this system in addition to contributing to the correct understanding of global warming, may also contribute to fundamental research on next generation resource utilization and environmental forecasting.