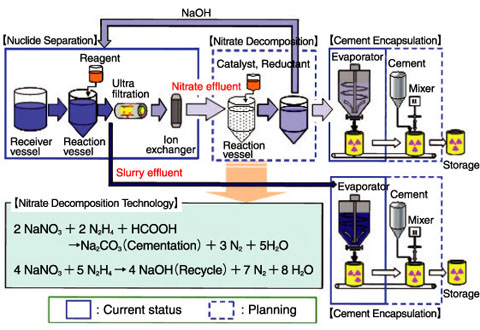
Fig.10-4 Current status and planning of the effluent treatment
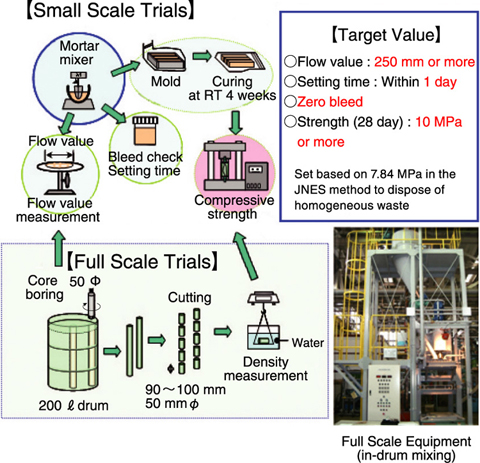
Fig.10-5 Diagram of cement based encapsulation trials
We are carrying out development work to install cement based encapsulation equipment at the Low-level radioactive Waste Treatment Facility (LWTF) (Fig.10-4). The effluent from the nuclide separation process is evaporated and encapsulated using an in-drum mixing technique. Because the ratio of water to salt is too high due to the efficiency of the evaporator, bleed water appears when commercial cement is used, and thus we adopted a special cement based on fine grains of blast furnace slag (BFS), which are capable of dealing with the surplus water. It has become possible to make a homogeneous waste form having strengths exceeding 10 MPa at a nitrate loading rate of 50 wt% (Fig.10-5). With regard to the slurry, it has become possible to make a homogeneous waste form by thickening the viscosity to prevent impurities from settling, and by adopting a paddle used for high viscosity fluids.
Carbonate effluent from the nitrate decomposition process mainly consists of carbonate precipitates and saturated solution of carbonate, having a pH of 11.5. Carbonates cause a shortage of water by hydrating 10 water molecules at temperatures under 32 °C and elevate the pH by reacting with calcium in a cement. We thus set the effluent temperature at 50 °C and adopted coarse BFS grains to deal with the shortage of water and the high pH. In small-scale trials, expansion was observed in some test pieces, and this seemed to be caused by decahydrate forming before setting, because of a rapid drop in temperature. In full-scale trials, however, such expansion in a test piece was not observed due to the retained heat in the 200 L drum. The obtained solid waste achieved homogeneity and compressive strengths exceeding 10 MPa at a carbonate loading rate of 30 wt%. Further trials are planned to obtain the conditions to inhibit the expansion.
Phosphate waste (NaH2PO4) is produced as a result of the treatment of solvent waste, and has a pH of 4. Therefore, it is necessary to neutralize the waste before mixing to increase the pH to make it compatible with the cement powder. The loading rate is lower than 6 wt% because the phosphate interferes with the hydration and its weight increases by the formation of 12-hydrate. To improve this situation, Ca(OH)2 has been used to convert the phosphate into stabile calcium phosphate having no hydrate. All the phosphate is converted into calcium phosphate, and the negative effect on hydration is eased, when the Ca/P molar ratio is more than 2.43. The waste form achieved homogeneity and compressive strengths exceeding 10 MPa at a NaH2PO4 loading rate of 14 wt%.
We will carry out trials to develop lower pH cement and to ascertain its suitability for disposal of waste forms.
<Previous: 10 Nuclear Fuel Cycle Technological Development | Next: 11 Development of Decommissioning and Radwaste Treatment Technology >