
Fig.9-6 Preparation method for reference material of solidified product containing α- and γ-ray emitting nuclides
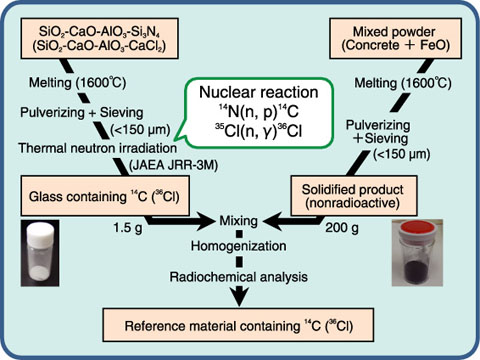
Fig.9-7 Preparation method for reference material of solidified product containing 14C or 36Cl
At the Advanced Volume Reduction Facilities (AVRF) of the JAEA, it has been decided that nonmetallic low-level radioactive solid wastes will be melted in a plasma heating furnace and converted to stable solidified products. For performing disposal of radioactive waste packages containing the solidified products, the evaluation of radioactivity inventories for selected nuclides is necessary. We therefore need to analyze radioactive samples similar to the solidified products to collect radioactivity data.
In order to verify the reliability of the radiochemical analysis, it is necessary to check whether an appropriate procedure has been followed. If we can use a reference material (RM) containing a known amount of the nuclides of interest, the reliability can be evaluated. In this study, preparation methods for simulated solidified products were investigated in order to prepare RMs for the radiochemical analysis of some α-, β- and γ-ray emitting nuclides.
At the AVRF, it has been decided that major non-metallic low-level radioactive solid wastes, namely concrete together with steel drams, will be processed by plasma melting. To simulate a major chemical composition of the solidified products, non-radioactive concretes and a chemical reagent (FeO) were used. In addition, we employed a laboratory-scale electric furnace to simplify the preparation method.
It is very difficult to ensure that volatile nuclides such as 14C, 36Cl, and 137Cs remain stable at high temperatures in a solidified product. It is well known that volatile metals such as Cs in the solidified products can be stabilized by maintaining low basicity. Under optimized conditions, it was found that solidified products containing 137Cs could be successfully prepared (Fig.9-6). For 14C and 36Cl, which are more easily volatile elements than Cs at a high temperature, it will be difficult to adjust the interest nuclide concentration in the solidified product on the preparation method of Fig.9-6. Therefore, we examined the following preparation method involving a nuclear reaction to produce a known amount of 14C or 36Cl in a glass. A RM containing either 14C or 36Cl was successfully prepared by the glass (1.5 g) and a solidified product (200 g) prepared from a mixture of nonradioactive concretes and FeO (Fig.9-7). We are planning to use the present preparation methods for the collection and evaluation of data on the radioactivity of wastes at the JAEA.
<Previous: 9-2 | Next: 10 Computational Science and E-Systems Research >