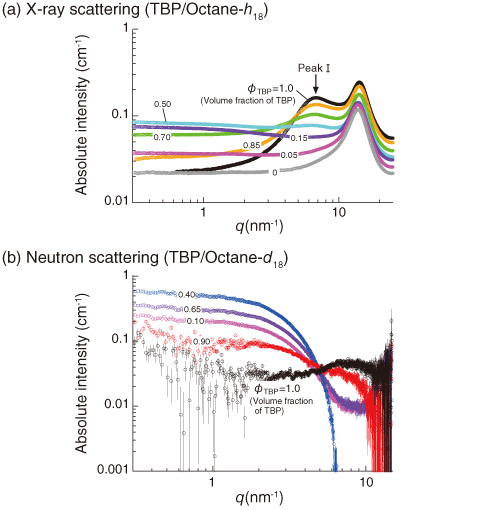
Fig.5-19 X-ray and neutron scattering profiles obtained for TBP/octane mixtures
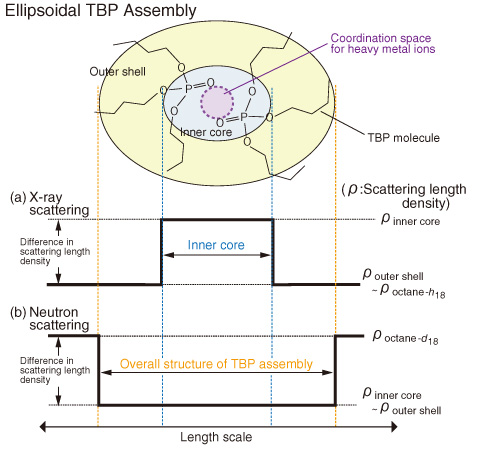
Fig.5-20 Diagram for scattering contrast and partial structure of ellipsoidal TBP assembly in X-ray and neutron scattering experiments
Tri-n-butyl phosphate (TBP) is an important extractant for separating hexavalent uranium, U(VI), and tetravalent plutonium, Pu(IV), from used nuclear fuel by solvent extraction. However, little was known on the structure and characteristic features of the TBP assembly and its coordination species with the heavy metal ions; thus, further study is needed to identify them. We aim to address these problems using scattering techniques; therefore, we collected X-ray and neutron scattering data for the TBP/octane mixtures as the first step of this study. As a result, we clarified the ordered structure formed by TBPs in octane.
In Fig.5-19(a), scattering maxima (peak I) due to the intermolecular interactions of two TBPs were clearly observed. It is evident that TBP forms a self-assembly in octane. In Fig.5-19(b), on the other hand, scattering from the overall structure of the assembly was observed at q < 6.0 nm-1. This indicates that TBPs form an ellipsoidal ordered structure, as illustrated in Fig.5-20, where the inside core is capable of incorporating a heavy metal ion.
The diagrams in Fig.5-20 indicate that (a) with X-rays the scattering length densities of the hydrophobic butyl groups in TBP (ρbutyl) and octane-h18 (ρoctane-h18) are roughly equal to each other, but that of the hydrophilic phosphate ester of TBP (ρhydrophilic) is larger than those of both hydrocarbon chains; and (b) with neutrons, the difference in the scattering length density between the hydrophobic and hydrophilic parts of TBP (ρbutyl and ρhydrophilic) is negligible as compared with the scattering length density of octane-d18 (ρoctane-d18). Note that X-rays and neutrons, respectively, enable the scattering to be effectively observed from the inner core of the TBP assembly and from its overall structure, respectively. In fact, the q dependence of the obtained scattering profiles differs greatly between X-rays and neutrons, elucidating the structural and characteristic features of TBP assemblies.
The quantum beam technique is a powerful tool for investigating the microscopic structures of the various organic molecules and their coordination species with heavy metal ions. Anticipated applications include the development of not only fuel reprocessing but also diverse research topics such as the recycling of valuable metals from the waste products or the polluted solutions.