
Fig.4-16 Flow electrolysis with column electrode
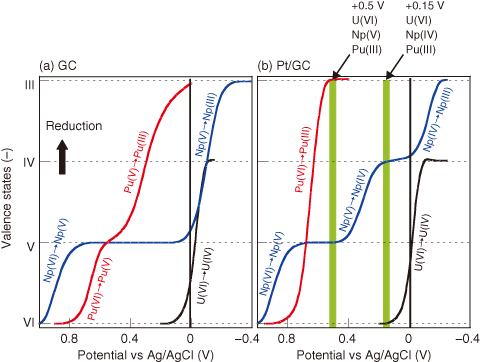
Fig.4-17 Valence changes of U, Np, and Pu ions by flow electrolysis
Actinides (An) such as uranium (U), neptunium (Np), and plutonium (Pu), in aqueous solutions exist as ionic species ranging from trivalent to hexavalent configuration. The chemical properties of these solutions depend on the valence of the ions; hence, a proper understanding of how to precisely adjust the valence of An ions in solution is required for efficient chemical separation and precise analysis.
We are developing a flow electrolysis method that uses as working electrode the column packed with glassy carbon (GC) fiber (Fig.4-16). Flow electrolysis is conducted by passing the solution through the column electrode, which can rapidly reach exhaustive electrolysis. The redox of An ions between tetravalent (An4+) and pentavalent (AnO2+) accompanies the transformation of ions. Consequently, the reaction is slow, and large overpotentials are necessary. We find that the overpotential decreases as a result of electrocatalysis when platinum (Pt) is used as the working electrode material. Reaction rates at normal Pt surfaces are very small, however, and a long time period is needed to reach exhaustive electrolysis.
To address this problem, we tried to develop a rapid electrolysis wherein the column electrode exploited electrocatalytic effect. A column electrode with the platinized GC (Pt/GC) fiber serving as electrode was prepared by electrodeposition of platinum black onto GC fiber from a chloroplatinic acid solution.
The relationships between the applied potential and the valence of U, Np, and Pu upon electrolytic reduction with GC and Pt/GC electrodes are shown in Fig.4-17. At the conventional GC electrode (a), Np(V) is immediately reduced to Np(III), whereas Pu(V) is slowly reduced to Pu(III) because of the large overpotential for Np(V) and Pu(V) reduction. These results indicate that the selective control of valence is difficult. However, at the Pt/GC electrode (b), Np(IV) can be precisely prepared. Thus, U, Np, and Pu ions can be prepared in various oxidation states (e.g., U(VI), Np(IV), and Pu(III) when +0.15 V is applied).
The results of this study may be applied to obtain precise coulometric speciation of individual valence ions by measuring electric charge during electrolysis. They may also be applied to controlling the valence for the separation of An ions.
The present study was partly supported by the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT) KAKENHI Grant-in-Aid for Scientific Research (C) (No.22560832).