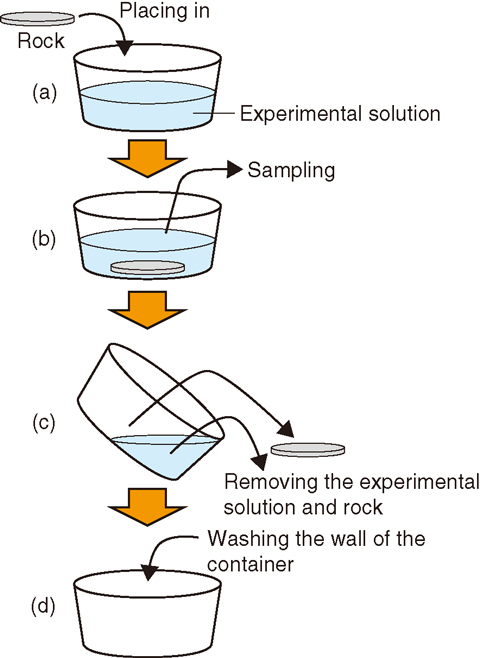
Fig.2-13 Flow of the sorption experiment
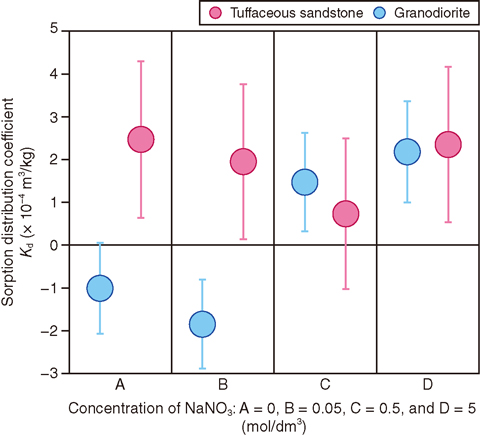
Fig.2-14 Sorption distribution coefficients of iodine on granodiorite and tuffaceous sandstone vs. concentration of NaNO3
Radioactive wastes generated from reprocessing spent fuel contain long-lived radionuclides such as iodine-129 (half-life: 1.6 × 107 year). If such wastes are disposed of into geological formations, nuclides may be eluted by groundwater and may migrate into the bedrock over a long period of time. The migration of nuclides will be delayed by the sorption of nuclides onto the bedrock. The sorption distribution coefficient, Kd, is used to evaluate this sorption.
Kd is the ratio of the element concentration in rocks to that in solution, and it never has negative values. It is difficult to directly and precisely measure the element concentration in the rock when Kd is obtained in experiments. The amount of the element sorbed onto the rock is thus determined as the difference between the element concentrations in the solution before and after contact with the rock, as shown in Fig.2-13. Either values with large errors or negative values should be obtained for the amount of the element in the rock because the measured elemental concentrations include errors due to the analysis and the handling. Such a problem is realized for low-sorptive elements (e.g., iodine) because the Kd is very small or close to zero. For such low-sorptive elements, it is essential to know whether even slight retardation of the migration in the bedrock due to sorption can be expected. It is thus necessary to clarify whether the obtained Kd values are significant by appropriately evaluating the errors.
In this study, sorption experiments of radioactive iodine onto tuffaceous sandstone and granodiorite were conducted for various sodium nitrate (NaNO3) concentration conditions by considering the effect of NaNO3 eluted from waste. The Kd was evaluated by subtracting the amount of iodine adsorbed onto the wall from the difference between the element concentrations in the solution before and after contact with the rock. Because the sorption of iodine onto rock was very small, various error propagations arising in the processes for the experimental solution were considered for the Kd evaluation. The experimental results showed significant values, which were non-zero under certain solution conditions, although the Kd of iodine was very small, as shown in Fig.2-14. The results showed the appropriate experimental and evaluation methods for the case where very small Kd values were expected for important nuclides involved in a safety assessment of waste disposal.
The present study was sponsored by the Secretariat of the Nuclear Regulation Authority (NRA) (formerly Japan Nuclear Energy Safety Organization, an incorporated administrative agency (JNES), formerly the Nuclear and Industrial Safety Agency (NISA), Ministry of Economy, Trade and Industry (METI), Japan).