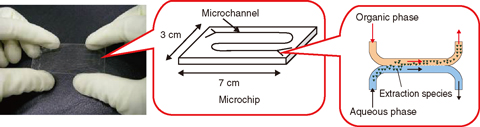
Fig.8-29 The glass microchip used for Pu extraction
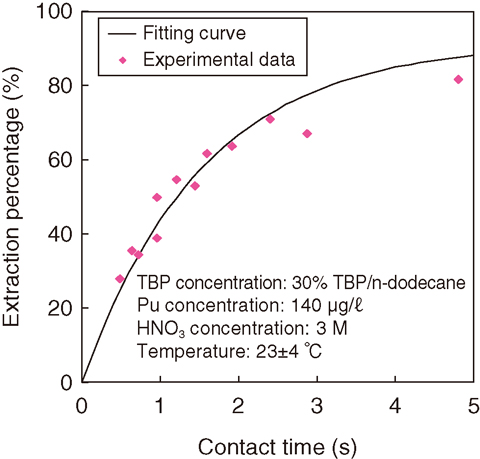
Fig.8-30 Dependence of Pu extraction on the contact time
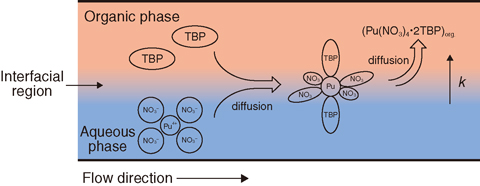
Fig.8-31 The model of Pu extraction in the microchip
The extraction of plutonium (Pu) by tri-n-butylphosphate (TBP) is used for spent nuclear fuel reprocessing. It is important to know the rate constant of this extraction system in order to design fast-extraction instruments such as centrifugal extractors. The single-drop method is primarily used to measure rate constants, although the specific interfacial area between the single drop and the surrounding liquid is generally small and limits the reaction. In this study, we used a microchip, which stably provides a large specific interfacial area, to evaluate the rate constant of Pu extraction.
The microchip, as shown in Fig.8-29, is a several cm square glass plate that typically has a few hundred μm channels. Effective extraction is expected in this micro-sized region due to a large interfacial area compared with the sample volume. Moreover, when the aqueous and organic phases contact in the microchip under a certain flow rate, a stable interface can be observed because of the laminar flow.
A solution of 3 M nitric acid including Pu and a solution of 30% TBP diluted with n-dodecane were used as aqueous and organic phases, respectively. These solutions were fed separately into the microchip and the interface between each phase was observed. It was found that a stable interface was formed for flow rates between 5 and 20 μℓ/min. The Pu extraction percentages at each contact time were obtained by analyzing the Pu concentration of the solution from the microchips. Fig.8-30 depicts the dependence of Pu extraction on the contact time with the time interval below 1 s. To evaluate the rate constant from this data, Pu extraction in microchip was modeled as shown in Fig.8-31. This extraction model was based on the Pu diffusion in the microchannel and the extraction at the interfacial region. A theoretical equation was derived from rate equations according to the model. It was fitted with the data in Fig.8-30 using the least-squares method. Consequently, the rate constant of Pu extraction was evaluated.
The results obtained in this study indicate that the microchip can be an effective experimental tool for studying the Pu rate constant. The data are expected to be used for the design of fast-extraction instruments. Furthermore, applications of microchips to the separation and analysis of radioactive materials are expected because microchip extraction can reduce the radioactive waste and downsize the analytical apparatus.
<Previous: 8-12 | Next: 9 Nuclear Fusion Research and Development >