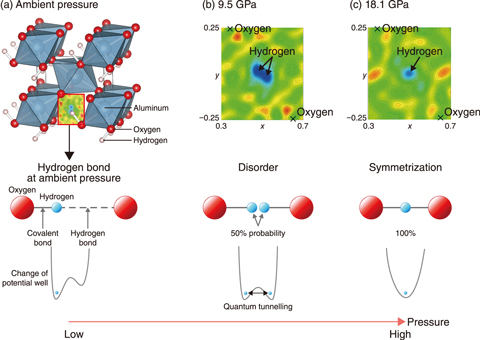
Fig.5-5 Crystal structure of δ-AlOOH at ambient pressure and change in the nuclear density map of δ-AlOOH at high pressure
Hydrogen exists not only on the Earth’s surface as liquid water but also as hydrous minerals in the deep mantle, which contain hydroxyl ions and/or water molecules in their structure. Since pressure and temperature increase as the depth increases in the Earth’s mantle, understanding the behavior of hydrogen bonds in minerals under such extreme conditions is necessary to understand the physical properties of the mantle.
On the surface of the Earth, hydrogen atoms generally take an asymmetric position between two oxygen atoms in minerals, forming a short covalent bond on one side and a long and relatively weak hydrogen bond on the other side. However, it was theoretically predicted in 1970’s that in high pressure ice, hydrogen is located at the center between two oxygen atoms in the so-called symmetrization of the hydrogen bond. Considerable effort has been expended to investigate pressure-induced hydrogen bond symmetrization, but most of the studies have been based on indirect methods such as X-rays and spectroscopy.
To investigate the effect of hydrogen bond symmetrization on physical properties of minerals, neutron diffraction experiments were conducted on δ-AlOOH (Fig.5-5(a)) using the high-pressure neutron diffractometer PLANET in the MLF at J-PARC. This hydrous phase is a potential candidate for the carrier of hydrogen into the deep mantle because of its wide stability field at high pressures and temperatures.
As a result, a decrease in the length of the hydrogen bond with the oxygen atom was observed at high pressure. Additionally, the strengthening of the hydrogen bond and weakening of the covalent bond were observed, as reflected by the decrease and increase in each distance, respectively. The hydrogen atom finally became equidistant between the two oxygen atoms at 18.1 GPa, which corresponds to the pressure at an approximate depth into the mantle of 520 km (Fig.5-5(c)). This result indicates that the hydrogen bond as an electrostatic interaction that is seen at the surface of the Earth disappears at the high pressure deep within the mantle. Instead, the hydrogen bonds covalently at high pressure with the oxygen atoms on either side. A disordered state was also observed below the symmetrization pressure, in which the hydrogen atom occupies two equivalent sites by quantum tunneling with 50% probability, as a precursor to the symmetrization (Fig.5-5(b)).
Previous studies have reported interesting changes to the physical properties in δ-AlOOH, such as a stiffening behavior along the directions where hydrogen bonds lie and an increase in the bulk modulus and sound velocities at high pressure. Furthermore, the conditions of these reported changes agree well with the detected pressures of the disorder and symmetrization. Using neutron diffraction, which allowed for the direct observation of hydrogen position, this work therefore revealed that a small change in the atomic position of hydrogen via the symmetrization can change the bulk physical properties of the mineral.
This work indicated that under high pressure conditions in the deep Earth, hydrogen bonds change in appearance and become more like covalent bonds due to the change in the distance between the oxygen atoms, increasing the elastic wave velocity. This detailed symmetrization of hydrogen bonds under high pressure must be considered when interpreting seismic wave velocity data based on the physical properties of hydrous minerals.
This study was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for Young Scientists (B) (No.22740349), and by Grants-in-Aid for Scientific Research on Innovative Areas (Research in a proposed research area) (No.20103001, No.15H05826).
(Asami Sano)