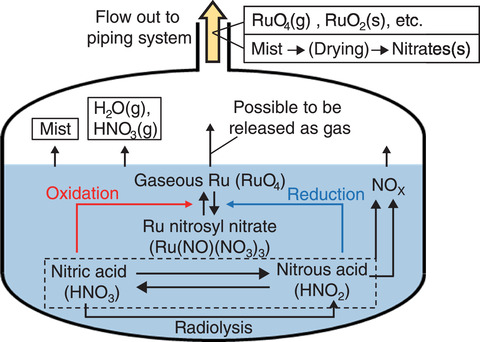
Fig.2-12 HLLW tank during an evaporation and dryness accident
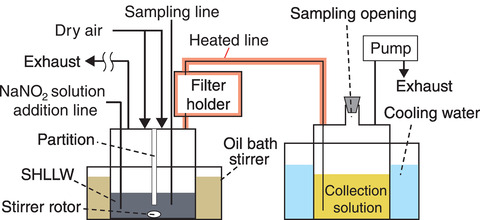
Fig.2-13 Experiment device for boiling SHLLW and collecting RuO4
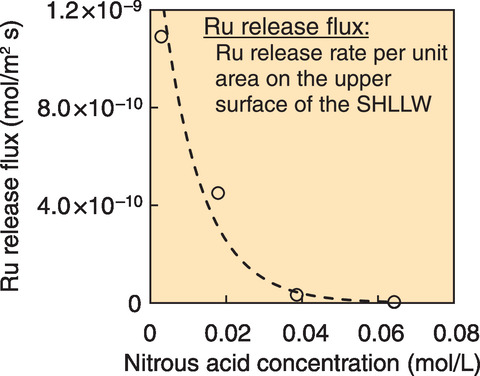
Fig.2-14 Relationship between nitrous acid concentration and Ru release flux
High-level liquid waste (HLLW) containing radioactive elements is temporarily stored in reprocessing plants, where uranium and plutonium are extracted from the spent nuclear fuels and the HLLW must be cooled continuously to remove decay heat. If the cooling functions are lost and countermeasures do not work sufficiently, the temperature of the HLLW may increase above the boiling point, the water will evaporate and the HLLW will dry up. This evaporation to dryness due to the loss of cooling functions has been defined as a severe accident (i.e., beyond the design basis) of the reprocessing plant; sufficient countermeasures to suppress the release of radioactive materials must be developed.
Since ruthenium (Ru) forms volatile compounds, such as ruthenium tetroxide (RuO4), more Ru would be released from the HLLW to out of the reprocessing plant than other elements in such an accident because RuO4 cannot be trapped in filters. Therefore, properly assessing the safety of a reprocessing plant requires an understanding of the release behavior of Ru.
RuO4 is generated by the oxidation of ruthenium nitrosyl nitrate by nitric acid in the HLLW. On the other hand, RuO4 can be reduced by nitrous acid, which is generated by the radiolysis of nitric acid in the HLLW, thereby reducing the release of RuO4 (Fig.2-12).
To clarify the relationship between the nitrous acid concentration and the amount of RuO4 released from boiling HLLW, simulated HLLW (SHLLW) prepared with non-radioactive compounds was heated under fixed conditions; the experimental design is summarized in Fig.2-13. For the nitrous acid concentration in the SHLLW keeping constant, sodium nitrite solution was continuously added. The amount of RuO4 released from the boiling SHLLW was trapped in the collection solution and measured.
As shown in Fig.2-14, increasing the nitrous acid concentration caused less RuO4 to be released. More specifically, increasing the nitrous acid concentration led to an exponential decrease in the Ru release flux. This result contributes to the quantitative analysis of released radioactive isotopes during reprocessing plant accidents.
This work was supported by the Nuclear Regulation Authority (NRA), Japan.
(Ryoichiro Yoshida)