Metallofullerenes are a kind of a clathrate compound which encapsulates metal atom(s) in a cage-like "Fullerene" molecule. It has been suggested that these molecules have characteristic electronic properties different from the empty Fullerene molecule as a result of the interaction between the encapsulated metal atom and the fullerene cage and, further, have uniquely characteristic chemical and physical properties. For these reasons, these molecules are noteworthy from the standpoint of developing new functional materials.
We have succeeded for the first time in synthesizing metallofullerene-encapsulated actinide atoms and in separating and purifying them selectively using High Performance Liquid Chromatography (HPLC), which uses the radiotracers 237U, 239Np and 240Am produced in the TANDEM accelerator.
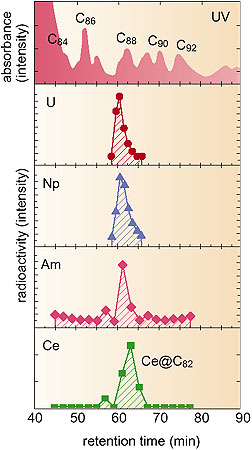
Figure 4-4 shows the HPLC elution behavior of U, Np and Am. For comparison, the elution behavior of metallofullerene-encapsulated Ce atoms is also shown in this figure. HPLC elution behavior is affected by the types of main products, the number of carbon atoms which constitute the fullerene cage, and the electronic state of the metallofullerene. Accordingly, the main species of the metallofullerenes of U, Np and Am are suggested to be M@C82 (the @ mark indicates the encapsulation host structure, where M@C82 indicates a Metal atom encapsulated in a C82 fullerene cage). This is similar to the Ce metallofullerene.
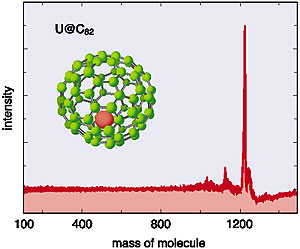
Next, we tried to produce and to isolate a macroscopic amount of U metallofullerene, and to investigate the properties of the main species of this metallofullerene using mass spectrometry, ultra-violet/visible/near infrared (UV/vis/NIR) absorption spectrometry, and x-ray absorption spectrometry. The main component of U metallofullerene was identified as U@C82 by mass spectrometry. Further, we have demonstrated the similarity of UV/vis/NIR spectra with those of M@C82 for most of the lanthanides such as (M=) La, Ce . . . etc. It is suggested that the electronic state of U@C82 is very nearly the same as that of M@C82 for the lanthanides, since the absorption spectrum reflects the both the energy differences and the transition rates between molecular orbitals. The oxidation state of the U atom in the fullerene is confirmed to be 3+. |