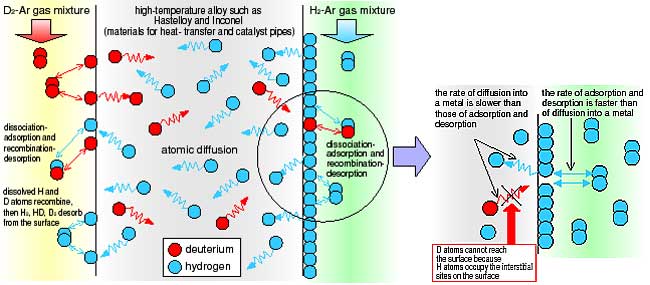
| Fig. 2-10 | Phenomenon of counter permeation of H2 and D2 Deuterium and hydrogen molecules will often dissociate and adsorb as atoms onto a metal surface. Some D and H atoms will recombine and desorb, while other atoms will diffuse into the metal. This diffusion will be in proportion to the concentration gradients of each type of atom. |
Fig. 2-11 | Blocking effect by dissolved H atoms on metal surface The rate of adsorption and desorption of H2 is higher than the rate of atomic diffusion of D. Thus, D atoms do not reach the metal surface due to the more active H2 adsorption and desorption on the surface. |
| Go back by your web browser, or click the right button. |
| Persistent Quest Research Activities 2002 Copyright(c) Japan Atomic Energy Research Institute |