Mösbauer spectroscopy, recoilless nuclear resonance absorption of gamma-rays,
is a powerful tool for probing the local electronic states around nuclei which
absorb gamma-rays, by measuring the energy shift of the absorbed gamma-rays.
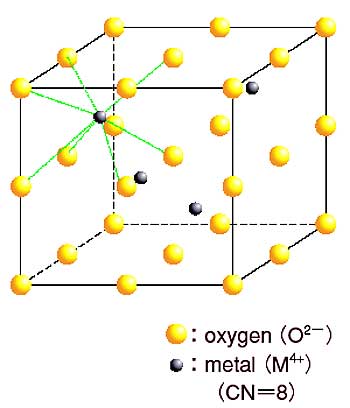
The local structure around Eu3+ in the fluorite-type oxides EuyM1-yO2-x (Fig. 6-3), (M=U, Th, Ce, Zr, Hf), was investigated by 151Eu Mösbauer spectroscopy over the whole range of 151Eu composition y.
Oxides LnyM1-yO2-x, in which tetravalent metal ions are substituted by trivalent
lanthanide cations (Ln3+), are ionic conductors owing to the high mobility of
the oxygen vacancies. The oxides with M=Ce, Zr, Hf, and Th have found application
to oxygen sensors, fuel cells, etc. The material properties of the nuclear fuel
oxides (M=U, Th) substituted by Ln, which is one of main fission products, are
important for investigating the irradiation behavior of nuclear fuels.
In this work, we have found that the isomer shift of the Eu3+ (peak of the Mösbauer
spectrum) depends on the coordination number of oxygen ions and on the Eu-O bond
length.
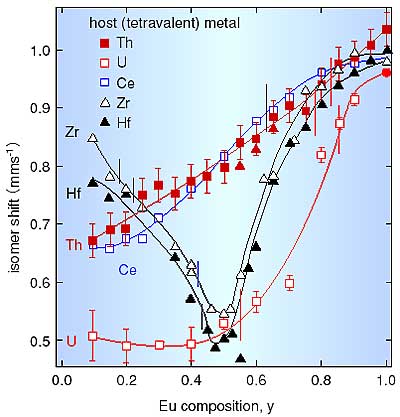
Fig. 6-4 shows the isomer shift dependence on Eu composition y. This dependence
reflects the different local structures in each system. For the M=Th, Ce systems,
an oxygen-deficient fluorite- type solid solution EuyM1-yO2-x (x=y/2) was formed, and the coordination number of oxygen ions decreased from 8 to 6.
For the M=Zr, Hf systems, the isomer shift has minimum at y=0.5, and the system
has an ordered pyrochlore structure in which the oxygen coordination number of
Eu is 8 and that of Zr and Hf is 6. In the composition range 0.2<y<0.45,
the Zr and Hf systems have fluorite-type structures from XRD measurements, but
the isomer shift dependence shows an inverse trend for the Th and Ce systems.
This result reveals that the Zr and Hf systems have a pyrochlore-type local structure
in this composition range.
For the M=U system in the composition range 0<y<0.5, a fraction of the tetravalent
U ions is oxidized to the pentavalent state in the substitution of U4+ by Eu3+.
An oxygen-stoichiometric fluorite-type phase (Euy3+Uy5+U1-2y4+O22-) is found to
be formed. Here, the oxygen coordination number is concluded to be 8, because
there are no oxygen vacancies. |