The energy spectrum of characteristic X-rays emitted from a material irradiated with ion beams reflects the electronic structure of the elements composing the material. This characteristic enables us to analyze trace amounts of elements included in the material with very high sensitivity in comparison with electron beam-induced X-rays. This method is well known as the name of "PIXE (Particle Induced X-ray Emission)" and has been applied to various fields of science and technology. In recent years, the PIXE method has become an effective new tool in medical and biological research for dealing with conflicting circumstances where, in some cases, the uptake of trace amounts of heavy elements in living organisms may be toxic for life conservation, whereas in other cases it may be essential for life conservation.
However, we cannot microscopically specify the location of heavy element uptake in living organisms using typical ion beams with the sizes of the order of mm. To make clear the effect of trace amounts of heavy elements on the biomechanism, ion microbeam is required to take trace-element mapping in the cells. We are now developing a micro-PIXE analysis system for elemental mapping under a joint research project between JAERI and the universities, using the JAERI light ion microbeams, which have the highest level of spatial resolution in the world.
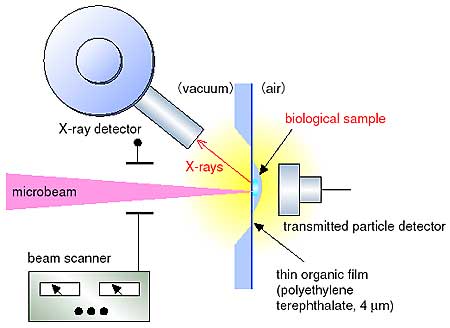
Some trace elements in biological samples may evaporate during analysis, because the samples are usually irradiated in vacuum. To minimize the evaporation, we have developed an in-air microanalysis system with a spatial resolution of 1 mm even after passing through a thin organic film which separates vacuum from atmospheric pressure. A multi-paramenter data acquisitions system has also been developed to detect X-rays and the beam scanning position on the sample simultaneously. On the basis of the above developments, we have constructed a two-dimensional imaging system for trace elemental mapping in a cell (Fig. 9-8).
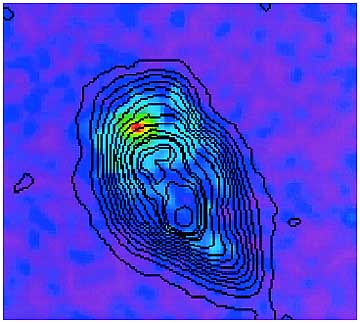
In a biological application of this system, we have succeeded in observing Fe accumulation in the cytoplasm. This suggests the presence of cytochrome C, the electron carrier in human leukemia cells, which is identified with programmed cell death (apoptosis) of 60Co gamma-rays iradiated cells (Fig. 9-9). This success has contributed to elucidation of the mechanism of radiation-induced apoptosis.
This technical progress for examining the kinetics of trace-elements in living cells will enable the investigation of cellular metabolism and the uptake of anticancer agents in biological cells. As a consequence, this system will be a useful tool for biological and medical research, and will offer the possibility of quantitative diagnoses in clinical applications. |