Weathering steel, steel that contains a small amount of Cr, Cu, and P, is widely used for structural purposes where it is exposed to the atmosphere and is not coated for protection against corrosives. It is recognized that the protective rust layer on weathering steel is responsible for atmospheric corrosion resistance because the microstructure of the rust layer, which consists primarily of goethite (alpha-FeOOH) containing Cr, is very fine with few cracks and pinholes and goethite, depending on the Cr content, has a selective permeability to restrict the permeation of corrosive anions. "Protect from Rust using its own Rust" - it is very important to clarify the role of additive elements for the corrosion resistance of weathering steel to develop new steels with superior performance.
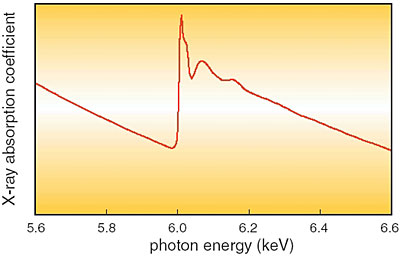
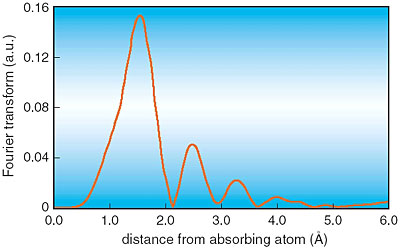
We have measured the energy dependence of the X-ray absorption coefficient, the so-called 'XAFS' spectrum (Fig. 5-15), using synchrotron radiation (SR) X-rays to investigate the chemical state of Cr in the rust layer of weathering steel. Analysis shows that Cr forming a chromium oxide ion is positioned at the vacancy site, not at the Fe lattice point, as a substitution in the goethite structure (Fig. 5-16). The negative charges of the chromium oxide ions restrict the penetration of corrosive anions, and these large ions disturb the atomic arrangement in goethite to make a fine microstructure.
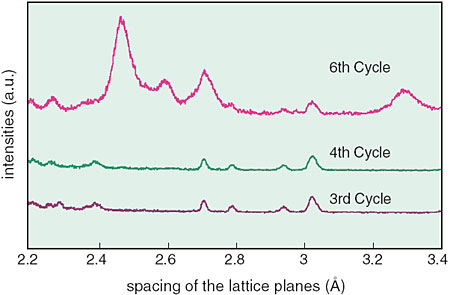
Also, we confirmed that Cr in a corrosive solution promotes the formation and growth of Cr-goethite on steel not containing Cr from an in situ observation of the corrosion process by SR X-ray diffraction (Fig. 5-17). This result suggests the possibility of the development of new corrosion protection techniques by surface treatments.
|