Cobalt (Co) atoms, having an electron-donor nature, exhibit important functions in the condensed phase as catalytic and magnetic substances. On the other hand, C60 cages are easily distorted by accepting electrons from bound partners, resulting in other carbon allotropes invaluable for nano-science and also for new, energy-related technologies.
In order to integrate the characteristics of Co and C60, we have explored the synthesis conditions for new structures with novel functions, starting from uniform mixtures. The mixtures were co-evaporated uniformly onto MgO.
Refined analyses with TEM and a Raman spectroscope reveal a "nano-composite structure," in which Co nano-particles with a metastable FCC structure are dispersed in a carbon allotrope matrix composed of polymerized C60, nanodiamonds, Single-Wall-Carbon-Nanotube (SWCNT)'s, and nano-graphites.
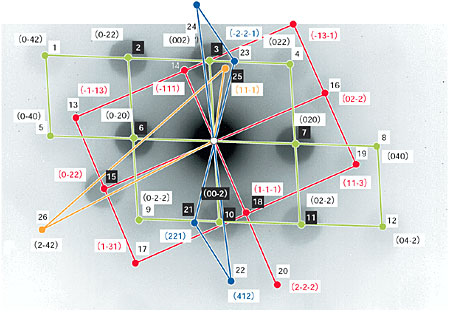
Fig. 7-6 shows diffraction spots from a selected area of the mixture. From a systematic analysis of the symmetry and spacing of the diffraction patterns, we have succeeded in concluding the following: 1) Co nano-particles have a metastable FCC structure different from the HCP structure of the bulk phase. 2) Nano-diamonds are hetero-epitaxially grown on Co nano-particles. 3) The nano-composite is pressurized by strain accumulated through Co precipitation.
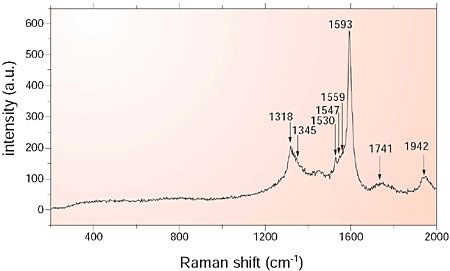
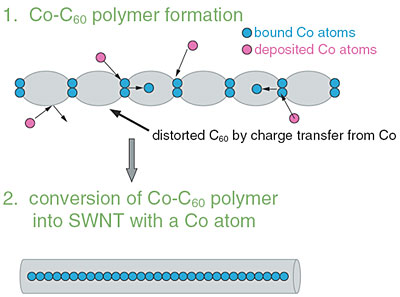
The polymerized C60 strings are easily converted into carbon nano-tubes by vacuum annealing. This process can be facilitated by small amounts of oxygen. A detailed Raman analysis was performed to elucidate the conversion process (Fig. 7-7). In the spectrum there remain small amounts of C60; however, one can observe the appearance of SWCT's clearly. The conversion can be understood reasonably by two sequential steps, C60 polymerization assisted by charge transfer from Co and a thermally-assisted change into carbon nano-tubes (Fig. 7-8).
These "nano-composites" can be assumed to be mixtures of hard nano-diamonds and soft carbon allotropes, and they have potential applications as solid lubricants for use in space under UHV conditions. On the other hand SWNT's with a Co string may find application as an elemental building block for magnetic devices in the future.
|