Since 1925, it has been well known that the addition of sulfur (S) to many metals and alloys causes them to become brittle. Metallic materials consist of crystal grains several tens of micrometers in size. The boundary between the crystal grains is called the "grain boundary". In the 1970s, it was found that S atoms gather at the grain boundaries by heating, etc., and thereby metallic materials easily break at the grain boundaries. However, it was not well understood why and how the S weakened the grain boundaries.
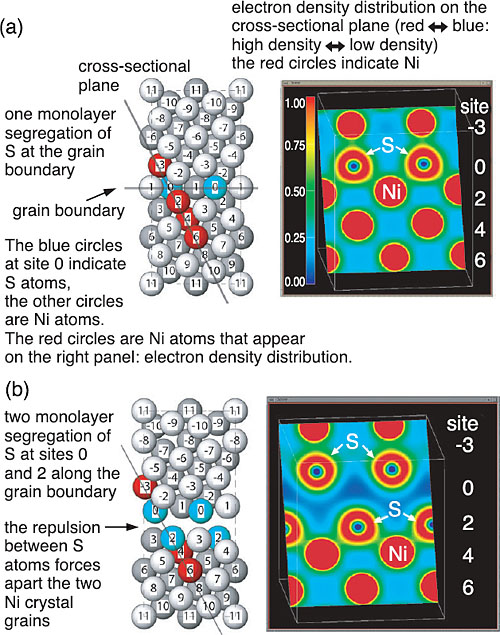
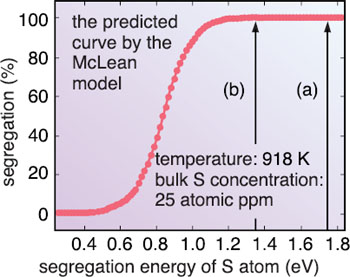
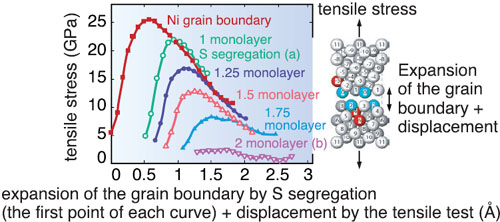
We simulated this phenomenon on a supercomputer using a first-principles calculation, which is the method to numerically solve the basic equation (Schröinger equation) of quantum mechanics and thereby clarify the properties of materials at the electronic level. We applied this method to a nickel (Ni) system; but similar results should be obtained for iron systems. First, we investigated the extent the S atoms can gather at the grain boundary by calculating the energy difference between the two cases; the S atom is in the inner bulk region and in the grain boundary (Fig. 10-2). As a result, we have shown that the S atoms can gather at the grain boundary even when the S concentration in inner bulk region is 25 atomic parts per million. Although this had been clarified by experiments, we determined that the S atoms can be adjacent to each other but then they cannot make strong bonds owing to the stronger bonds between Ni and S atoms. This leads to a long S-S bonding distance that forces apart the two Ni crystal grains (Fig. 10-1). From first-principles tensile test calculations, we found that the maximum tensile strength is reduced by one order of magnitude by the repulsive interactions among S atoms (Fig. 10-3). Thus, we have determined that the repulsive interactions between segregated S atoms cause metals to become brittle. |