In the course of investigating the behavior of actinides in the
natural environment and developing the separation of actinides,
information on their chemical state is necessary. We have intensively
examined a reliable and sensitive method for the detection and
speciation of actinide species in solution with the aid of a continuously
variable wavelength dye-laser which can selectively excite a chemical
species.
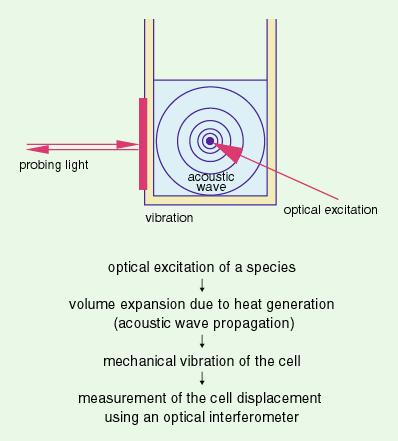
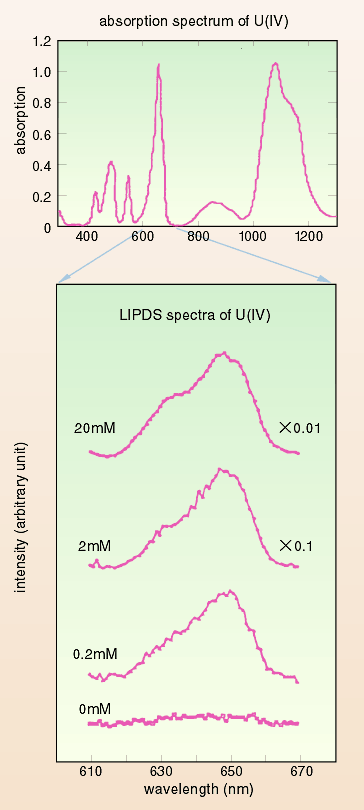
Laser induced photothermal displacement spectroscopy (LIPDS) is
based on selective optical absorption by actinides in solution,
followed by the relaxation of this absorbed (electronically excited)
energy into heat and an acoustic wave thereby produced which causes
vibration of the sample cell and thus can be detected. A variety
of potential applications comes from the remote and non-contact
detection of the mechanical vibration of the cell.
Furthermore, electrons in the f-orbitals in many lanthanides and
actinides may be excited and de-excited with emission at wavelengths
a little different from that of the excitation. The excitation
energy in this case easily goes to the energy of O-H vibration
of the water molecules bound directly to the ion, the lifetime
of the fluorescence being dependent on the number of water molecules
in the hydration shell. With the aid of this relationship one
can get information on the chemical state such as the number of
water molecules around a metal ion in solution, in solid, or at
their interface. |