Recently, an attempt of the high burnup of fuel has been performed
due to the demand for the cheaper cost of electricity generated
by nuclear energy. The fission products and transuranium elements
(Np, Pu, etc.) are accumulated in the oxide fuel with high burnup.
It is very important to study the vaporization behavior in the
complex oxides of perovskite type such as SrPuO3, in order to examine the safety of fuels under long time irradiation
and high temperature conditions.
Study of the thermodynamic properties of the complex oxides containing
Sr, Ba, etc. (alkali earth elements) and U, Np and Pu (actinide
elements) has been carried out in collaboration with the University
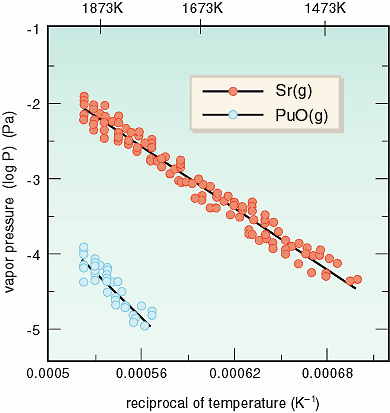
of Tokyo. The thermodynamic quantities of the vapor pressures
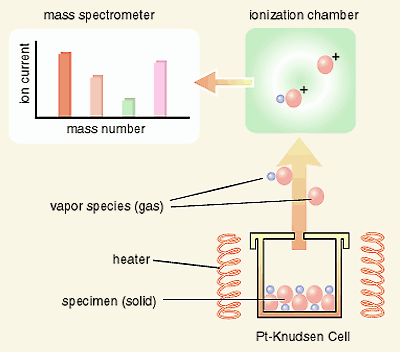
of SrPuO3 and the standard molar enthalpy of formation of SrPuO3 have been measured using a quadrupole mass-spectrometer equipped
with a Knudsen-cell as shown in Fig. 3-3, which is installed in
a plutonium glove box maintained at negative pressure with respect
to the atmosphere, as shown in Fig. 3-4. In addition, the mechanism
of vaporization and decomposition has been clarified by analyzing
the vaporized species over SrPuO3. At present the study of the vaporization behavior of BaPuO3 is being performed. |