How do you imagine the microstructure of fission-produced tervalent
lanthanides and actinides in nitric acid solution during nuclear
fuel reprocessing? We obtained information on the chemical species
of aqueous lanthanum ion in solution systems by 139La nuclear magnetic resonance (139La NMR). NMR is a magnetic resonance phenomenon of the central
nucleus of an atom, as it is, and it reflects the electronic symmetry
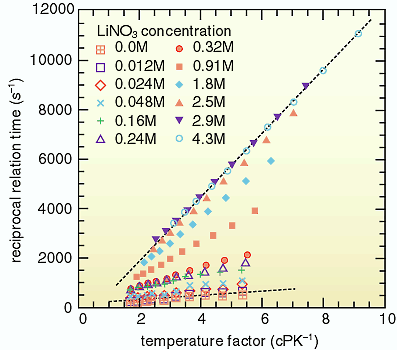
surrounding the nucleus. One of the experimental results, Fig.
3-7, shows the relation between the temperature factor and the
reciprocal of longitudinal relaxation time (T1, the recovering period to the initial state from the radiowave-induced
resonance state). The relaxation time is determined by the electronic
structure surrounding the nucleus as mentioned above, and the
mobility of the ion in the solution system. As the ion mobility
is changed with temperature, the slopes of the straight lines
in Fig. 3-7 indicate the symmetry of the electrons surrounding
the nucleus. Relaxation occurs slower in a state of higher symmetry
of electrons surrounding the nucleus and in such a more symmetric
structure, the slope is lower. Conversely high slope indicates
lower symmetry. From Fig. 3-7, it can be seen that the slope of
the straight line becomes steeper with increasing concentration
of nitrate solution.
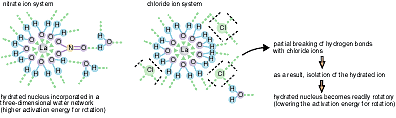
Considering the activation energies of rotation in the nitrate
and chloride ion solution system (obtained by the NMR method),
together with the results in Fig. 3-7, it is shown that, as in
Fig. 3-8 La3+ forms LaNO32+ in 0.05-3 M nitrate solution, while, in the chloride solution,
La3+ is directly surrounded by no chloride ions but by water molecules.
Furthermore, over 0.6 mol/l chloride solution, the hydrated La ion becomes isolated from
the three-dimensional water network by the effect of the chloride
ions breaking the network. The NMR method allows deeper understanding
of the solution systems and is a way to understand the process
chemistry and is useful for the safe and high performance of reprocessing
as well. |