The synthesis of a single crystal is essential to study the fundamental
properties of materials. Ordinarily, many single crystals are
grown in thermal equilibrium, but this thermal equilibrium method
is inapplicable to some crystals. One example of this inapplicability
is the synthesis of a zirconium dioxide (ZrO2) single crystal, which is used for a crucible at high temperature
and for a film used for the separation of hydrogen gas. This crystal
transforms into three different crystal structures, cubic, tetragonal
and monoclinic structures, at high temperature. Since the transformation
from the tetragonal to monoclinic structure at 1,170 degrees is accompanied by a large and destructive volume change,
the synthesis of a stable single crystal with monoclinic structure
by the thermal equilibrium method has been impossible at room
temperature.
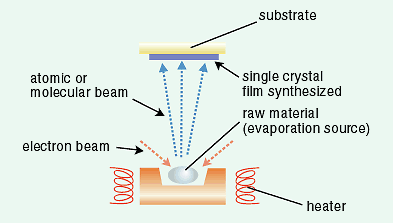
In this study, we were able to synthesize a ZrO2 single crystal film on a sapphire substrate by using the molecular
beam epitaxy method as shown in Fig. 3-11. The structure of the
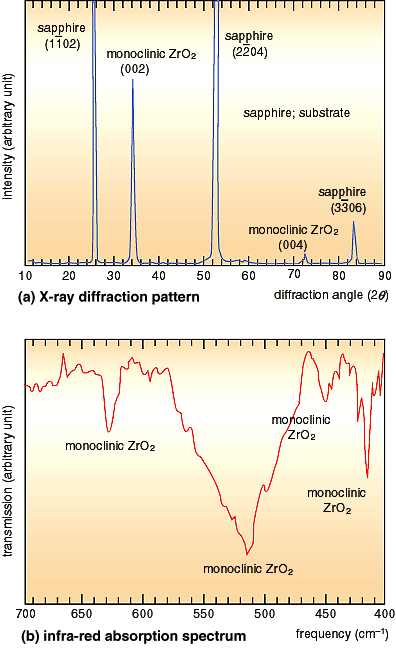
film examined by X-rays and infra-red spectroscopy is shown in
Fig. 3-12. A good quality and homogeneous single crystal film
is demonstrated by the sharp diffraction peaks and diffraction
angles of the X-rays, and also the transmission frequencies in
infra-red absorption. This molecular beam epitaxy method enabled
us to synthesize new single crystals such as strontium oxide (SrO)
which has been impossible to synthesize by the thermal equilibrium
method. With this new method of synthesizing crystals, we can
exactly measure some properties of the materials such as ionic
conductivity which cannot be obtained for polycrystalline solids. |