Publication Date: March 12, 2026
Access counts:0
Enthalpy Measurement Approaching 2000 ℃
-Study on Advancement of MOX Temperature Analysis-
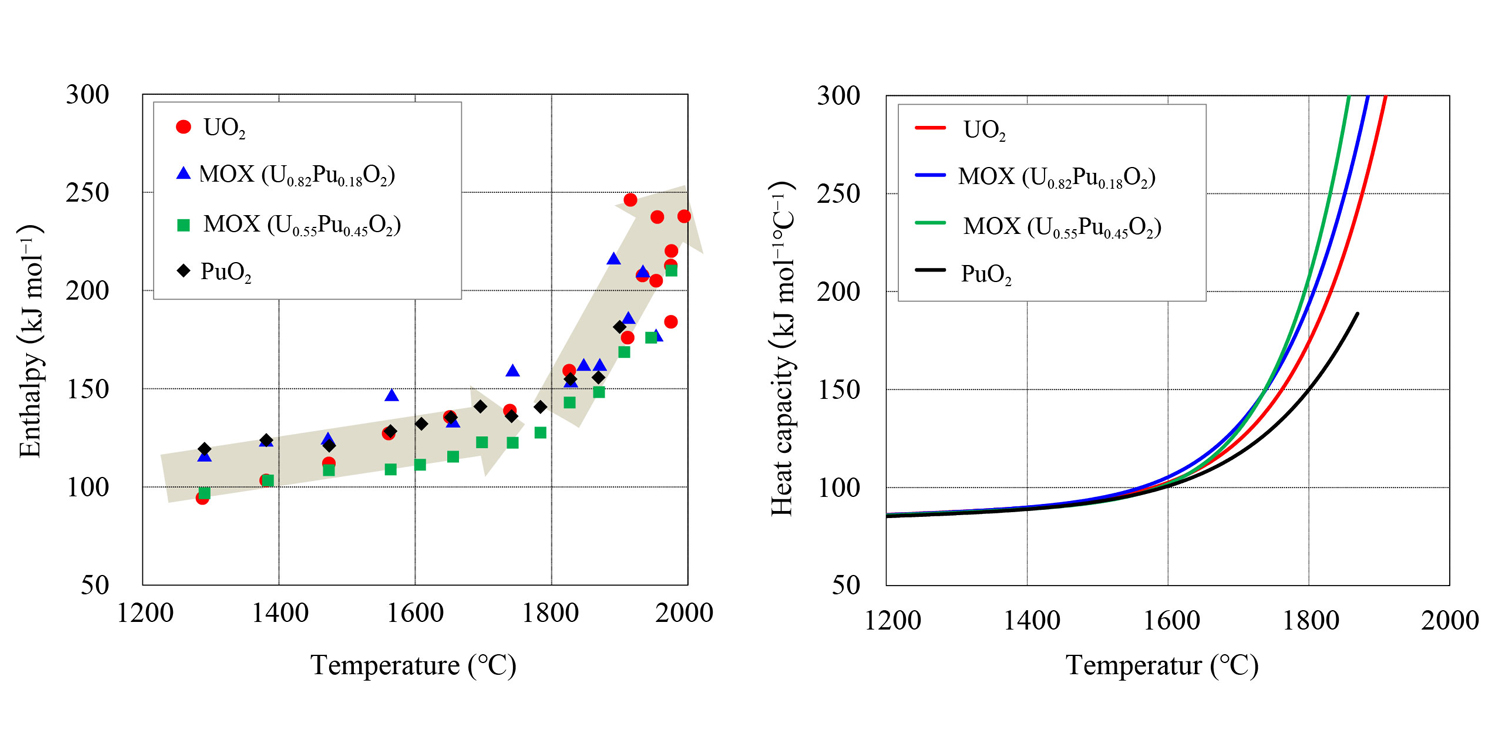
Fig. 1 Measured enthalpy (left) and evaluated heat capacity (right) on UO2, MOX, and PuO2
To perform temperature analysis on uranium-plutonium mixed oxide (MOX) fuel, accurate understanding of the fuel properties is required. The MOX fuel pellet reaches high temperatures exceeding 2000 ℃; however, its fuel properties at such high temperatures are still not known with sufficient accuracy. Therefore, it is important to expand such property data, clarify the underlying mechanism, and improve the reliability of the properties. In this study, we measured the enthalpy of MOX, UO2, and PuO2 at temperatures approaching 2000 ℃, which is experimentally achievable, and determined the heat capacity from those measurements.
The measured enthalpy data, along with the analysis results of heat capacity determined from those enthalpy values, are shown in Fig. 1. For all uranium-plutonium compositions, a steep increase in enthalpy was observed starting around 1800 ℃. The results indicate that composition of the fuel itself has little effect on this trend. Heat capacity is obtained as a temperature derivative of enthalpy. Consistent with the enthalpy trend, heat capacity also showed a steep increase as the very high temperatures are reached.
Concerning the experimental data for enthalpy at temperatures approaching 2000 ℃, this study is the first to experimentally compare MOX, UO2, and PuO2. The data obtained in this study can contribute to more accurate temperature analysis on MOX fuel.
If you have any comments or feedback about this page, please click the button to share your thoughts with us.
