Publication Date: March 31, 2026
Access counts:0
Achieving High-Density Heat Storage Through the Nanosheet Formation of Layered Mn Oxides
-Contributing to Carbon Neutrality Through Recovery of Unused Low-Temperature Waste Heat-
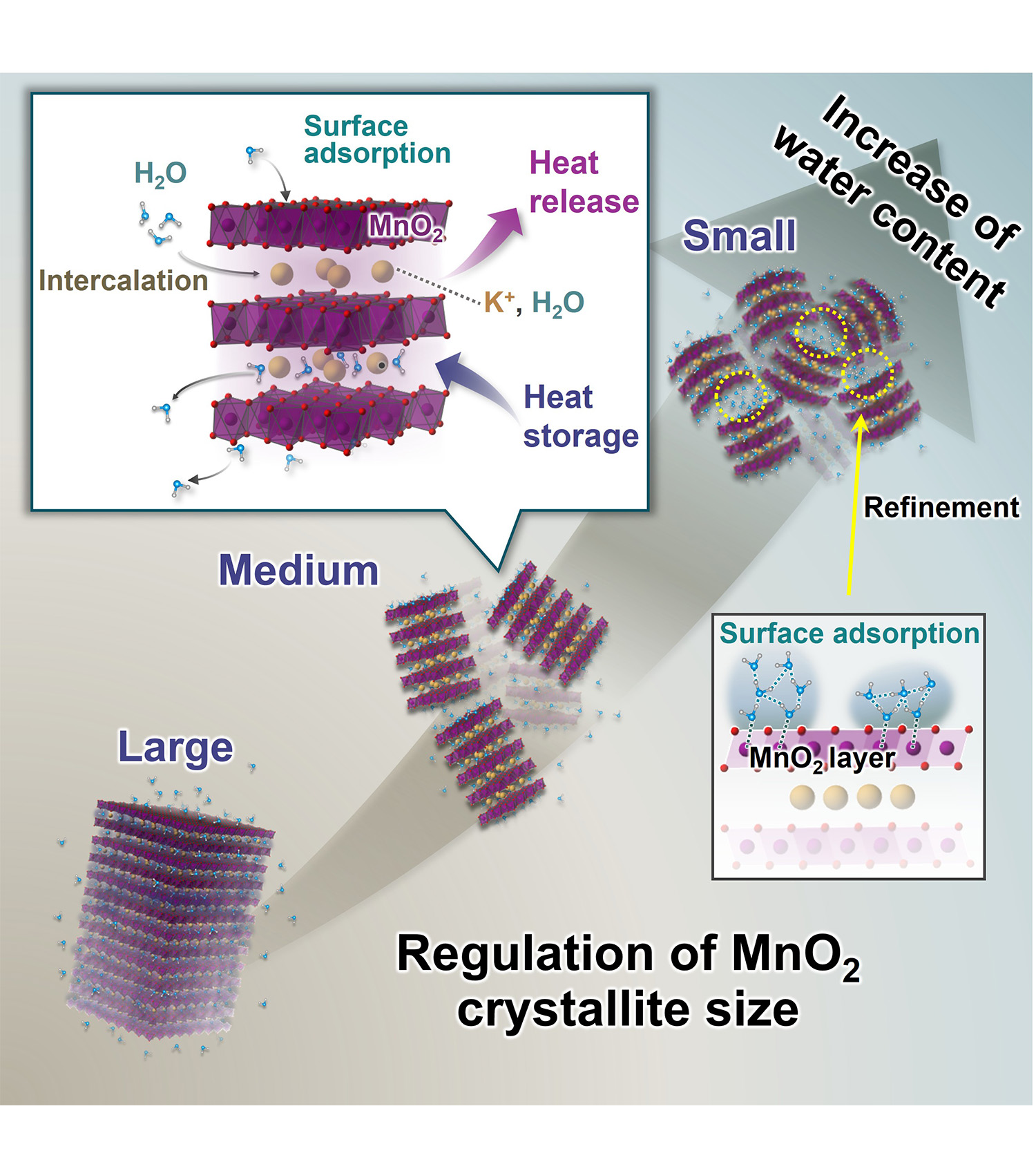
Fig. 1 Dual heat storage mechanism via intercalation and surface adsorption
Low-temperature waste heat below 200 ℃ occupies two-thirds of industrial waste heat. Such low-temperature waste heat should be recovered and reused for achieving carbon neutrality. Layered manganese dioxide has been found to release interlayer water molecules at approximately 130 ℃ and to absorb moisture from the atmosphere at room temperature, releasing heat in the process. This property indicates that layered manganese dioxide can be a material capable of high-density, high-speed heat storage and release.
In this study, we found a second mechanism, surface adsorption emerging at temperatures below 60 ℃ by breaking down layered manganese dioxide into ultrathin nanosheets (Fig. 1). This is attributed to surface water molecules trapped in pores of manganese dioxide nanosheets. The dual mechanism via intercalation and surface adsorption increased the total amount of storable water molecules by 1.5 times and enhanced the energy storage density by approximately 30 % when compared to bulk MnO2. Furthermore, the nanosheets can effectively operate at much lower temperatures.
This achievement demonstrates the potential for heat storage at even lower temperatures than previously possible. The superior heat storage performance of the layered manganese dioxide is expected to be applied to diverse energy-saving technologies, such as nighttime heating using daytime solar heat, warming-up operation, and offline waste heat transport.
The achievement of this study has been published in the journal Communications Chemistry on June 3, 2025.
If you have any comments or feedback about this page, please click the button to share your thoughts with us.
