Publication Date: April 17, 2026
Access counts:0
Effect of Temperature and Shear on Plant Protein Structure
-Molecular Mechanisms Underlying the Texturization of Meat Analogs-
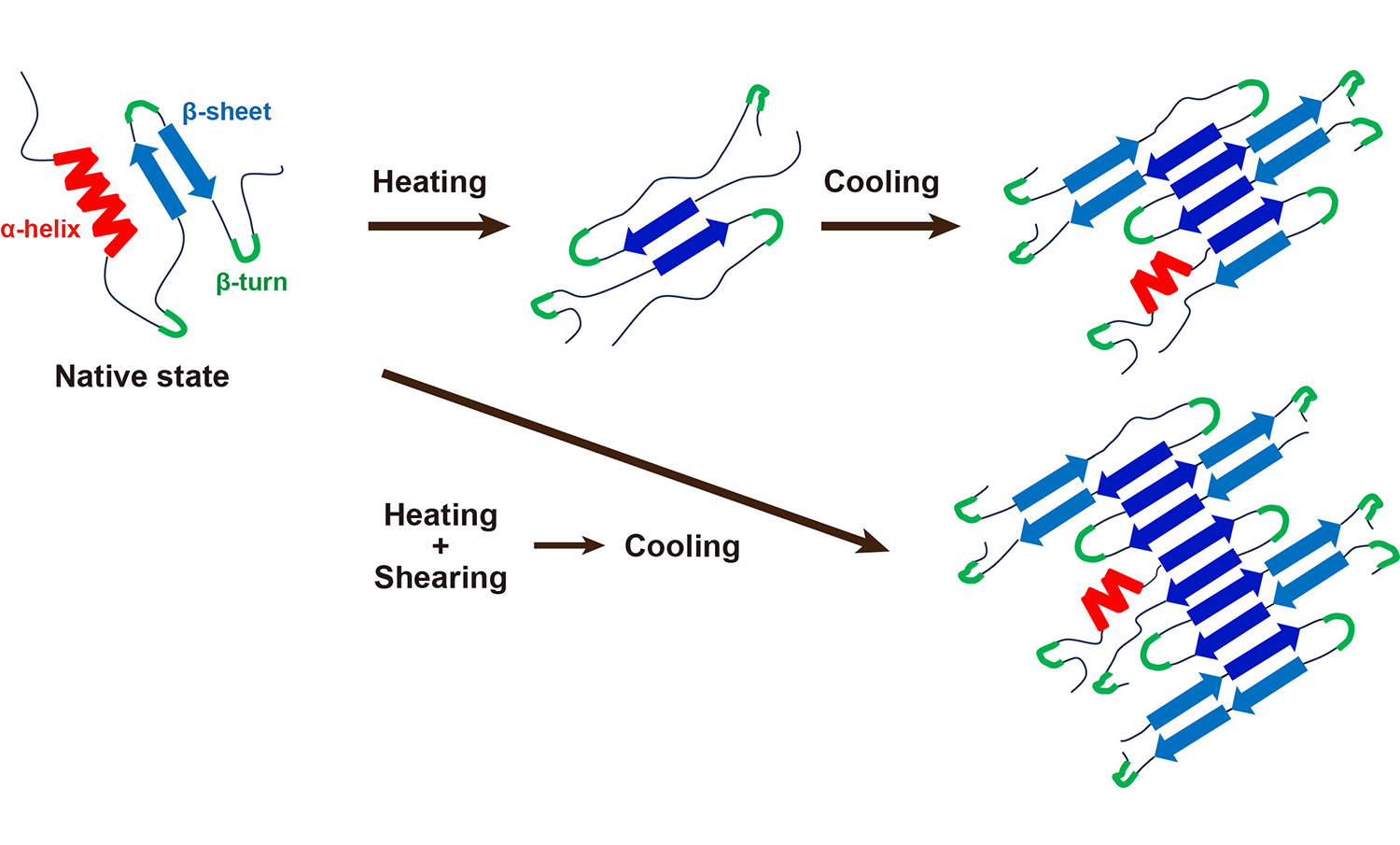
Fig. 1 Schematic illustration of structural changes in plant proteins induced by heating, shearing, and cooling
The structuring of plant proteins by extrusion into texturized fibrous matrixes that confer a meat-like texture is widely employed as the primary process in the production of meat analogs. However, the complex interactions within an extruder—in which temperature, water content, and shear act simultaneously—have hindered a fundamental understanding of how individual processing parameters transform protein molecular structure and ultimately determine the texture of the final product. In particular, it remains challenging to disentangle the respective contributions of heating, cooling, and shearing at the level of protein secondary structure.
In this study, we focused on protein secondary structure and investigated the effects of heating, cooling, and shearing on pea protein isolate. Temperature-controlled attenuated total reflectance–Fourier transform infrared spectroscopy was employed to monitor the heating–cooling cycles, while the combined effects of heating and shearing were evaluated via microcompounding, which closely mimics extrusion at the gram scale. Upon heating in the absence of shear, native α-helixes and intramolecular β-sheets unfolded into random structures, followed by the formation of intermolecular β-sheets that induced aggregation. During subsequent cooling, these intermolecular β-sheets became increasingly ordered and extensive, thereby strengthening the network. Concurrently, portions of the random coil domains partially refolded into non-native β-structures (Fig. 1, top). In contrast, when shear was applied at elevated temperatures, β-sheet formation became more extensive, resulting in β-rich structures dominated by intermolecular β-sheets (Fig. 1, bottom). Under these conditions, an entangled network of protein chains developed, yielding a cohesive protein matrix with a chewy texture.
For the first time, this study elucidates protein structural changes during extrusion processing at the secondary structure level. These molecular-level insights into plant protein texturization are expected to facilitate more precise control in food processing design for meat analogs and a broad range of functional food ingredients.
If you have any comments or feedback about this page, please click the button to share your thoughts with us.
