Publication Date: March 6, 2026
Access counts:0
Rapid Removal of Radioactive Cesium from Soil Using the Power of "Salt" × "Vacuum"
-A New Decontamination Mechanism Driven by Vacuum-
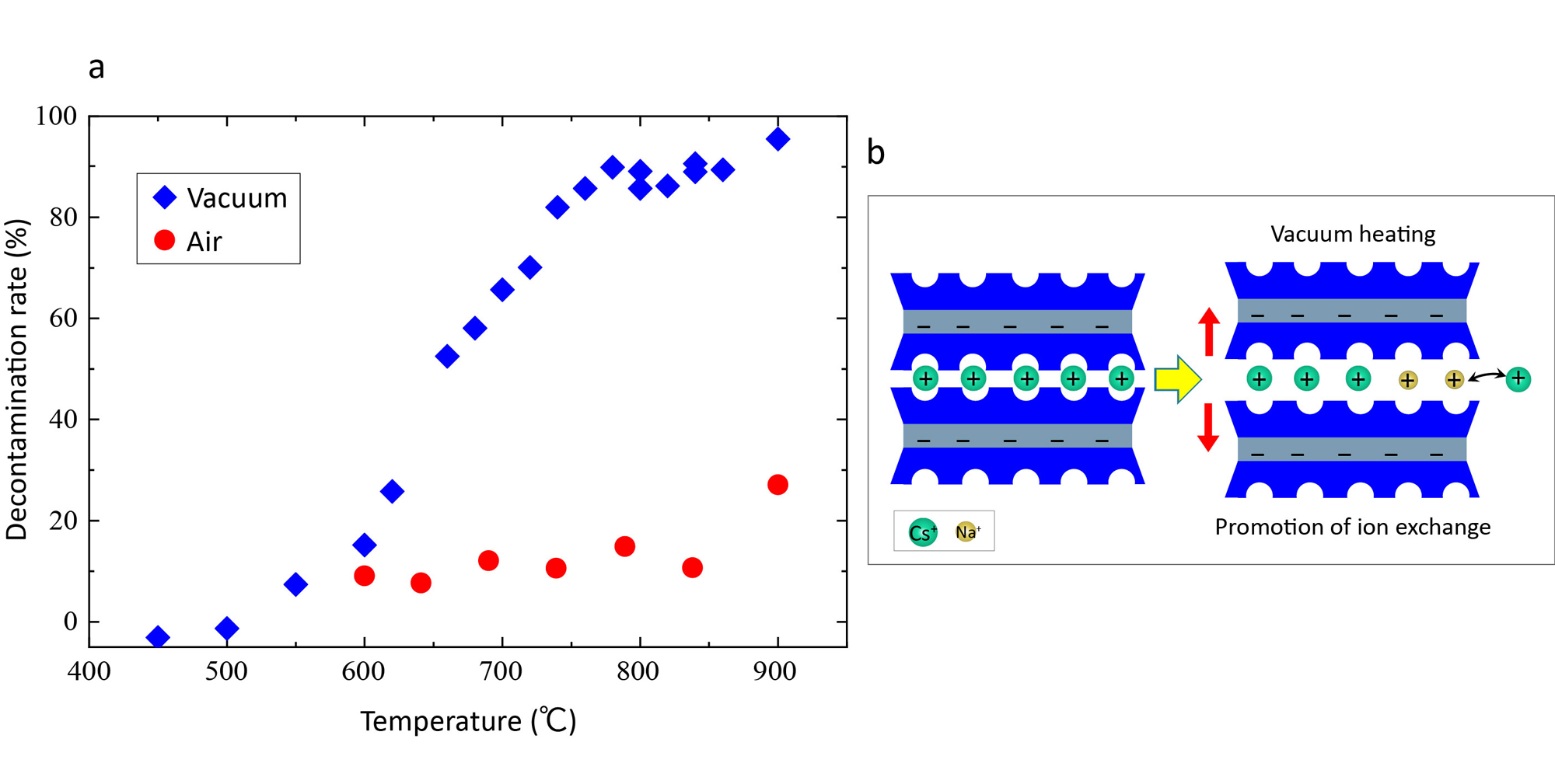
Fig. 1 (a) Comparison of decontamination rate in vacuum and air conditions
and (b) schematic of vacuum-promoted Na+ ![]() ↔ Cs+
↔ Cs+ ![]() ion-exchange mechanism
ion-exchange mechanism
Researchers led by Iwao Shimoyama at the Advanced Basic Research Center have demonstrated that vacuum heat treatment using sodium chloride (NaCl) enables highly efficient removal of radioactive cesium (Cs) at significantly lower temperatures than conventional thermal treatments, which typically require 1000–1300 ℃. When NaCl was added to contaminated soil at a 1:1 weight ratio and heated at 800 ℃ for 2 hours under vacuum, cesium removal was approximately six times more efficient than under identical conditions in air, achieving a decontamination rate exceeding 90 % (Fig. 1a, b).
Under vacuum, the sublimation of NaCl is promoted, supplying sodium ions to the interlayers of clay minerals. This expands the interlayer spacing and drives a rapid ion-exchange reaction in which cesium ions are rapidly replaced by sodium ions. As a result, an ion-exchange process that typically requires long reaction times in aqueous methods was completed in just one hour from the start of heating.
This achievement enables low-temperature and energy-efficient thermal decontamination, opening a pathway to the effective volume reduction and resource recycling of radiologically contaminated soils. The findings were published online in the Journal of Environmental Management on June 19, 2025.
This work was supported by JSPS KAKENHI Grant Number JP16H02437.
If you have any comments or feedback about this page, please click the button to share your thoughts with us.
