Fig.14-7 Outline of main pyro-reprocessing steps
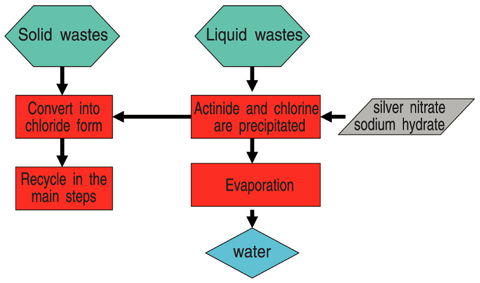
Fig.14-8 Flow chart of waste processing
The pyro-reprocessing (Fig.14-7) is one of the advanced technologies for the separation of actinide elements from spent nuclear fuel. The elements are electrochemically separated in a molten salt medium unlike the conventional aqueous-reprocessing, which is basically performed in nitric acid and an organic solvent medium. Since actinide elements are naturally recovered together in the pyro-reprocessing, this process has a superior resistance to the nuclear proliferation. Moreover, this process is expected to enhance the economical potential of nuclear fuel reprocessing, because the process itself is quite simple when compared to the conventional aqueous-reprocessing. Concerning the wastes from the pyro-reprocessing, some of the waste might contain significant amounts of actinide elements. It is believed that the recovery of these actinide elements enhances the potential of the pyro-reprocessing in regards to the efficient utilization of nuclear resources and reduction of environmental impact.
The actinide in the chemical formation such as oxide, which can not be treated in a normal process (Fig.14-7), is included in the solid wastes of the residue from the electrodes and sediments. On the other hand, the process operation of the pyro-reprocessing requires a chemical analysis in each step in order to control the process condition. Various liquid wastes containing actinide elements are generated from the analysis. A procedure is being developed to recycle the actinide elements from these wastes.
By conversion of the solid wastes to a chloride form, the actinide elements in the solid wastes can be put back into the pyro-reprocessing. The solid wastes was poured into a LiCl-KCl molten salt, which is the same mixture as the agents currently used in the electrolytic refining process, and a chlorination reagent (zirconium chloride) was gradually added to the LiCl-KCl molten salt. Most of actinide elements were successfully converted to a chloride form after sufficient reaction time (about 10 hours). As for the liquid wastes, the separation method by evaporation was investigated. The actinide elements were recovered as a residue. Since the liquid wastes contain chlorine which causes corrosion of equipments and harmful cadmium, it is necessary to prevent these substances from converting to water which is separated from the residue. These elements are removed by precipitation by using silver nitrate and sodium hydrate before the evaporation. The actinide elements in the residue are also converted to a chloride form as mentioned above.
We have not only been studying the main steps of pyro-reprocessing but also, the incidental technology such as waste treatment (Fig.14-8), in order to realize the pyro-reprocessing for the advanced nuclear reactor fuel. This study is being performed in the Chemical Processing Facility (CPF) in collaboration with Central Research Institute of the Electric Power Industry.