Fig.6-6 17O-NMR study of UO2, NpO2 and PuO2
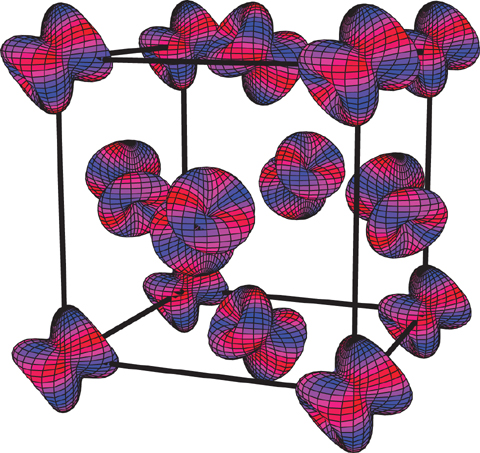
Fig.6-7 Magnetic octupolar ordering in NpO2
To understand macroscopic properties of materials, it is essential to understand their electronic properties from a microscopic viewpoint. Recently, we have carried out a series of nuclear magnetic resonance (NMR) studies for actinide oxides ( AnO2 : An= U, Np, Pu, Am, etc ). The microscopic NMR data make a clear distinction between their electronic properties at low temperatures.
Fig.6-6 shows the 17O-NMR spectra observed in UO2 , NpO2 and PuO2 , respectively. There is a marked difference in the width and shape of the spectra. For UO2 (a), we have obtained a broad spectrum with a rectangular line shape. This characteristic line shape indicates the existence of a large internal filed at oxygen sites due to the ordering of magnetic dipoles. On the other hand, the spectrum for NpO2 (b) broadens rather moderately, as compared with that for UO2, while it exhibits a complex structure. The electronic ground state of NpO2 had remained a mystery for many years. Our recent NMR study on a single crystal has revealed the occurrence of a novel magnetic octupolar ordering in the ground state (Fig.6-7). Finally, we have observed a narrow spectrum for PuO2 (c). This confirms that a nonmagnetic electronic ground state is realized even at the low temperature of 6K.
The actinide dioxides discussed here are all insulators with the same cubic crystal structure. Even though, these materials exhibit a variety of electronic states at low temperatures. Why they are so different?
All the actinide ions in AnO2 have the same tetravalent state. Therefore, the number of localized f-electrons per ion is two for U4+, three for Np4+ and four for Pu4+. In addition, due to the strong spin-orbit interaction, the f-electrons carry multipole degrees of freedom: dipole, quadrupole, octupole, etc. The available multipoles on each material are dependent on the number of the f-electrons as well as the symmetry of crystal. A variety of physical phenomena result from a variety of available multipoles in AnO2 .
AnO2 represents perhaps the most studied series of any actinide compounds. From a chemical and industrial perspectives, this interest has stemmed from their use as nuclear fuels. Nowadays, however, AnO2 is also an important series of materials to promote a better understanding of the fundamental physics of multipoles. We are now preparing to advance our NMR study to AmO2 as a joint work with Tohoku University.