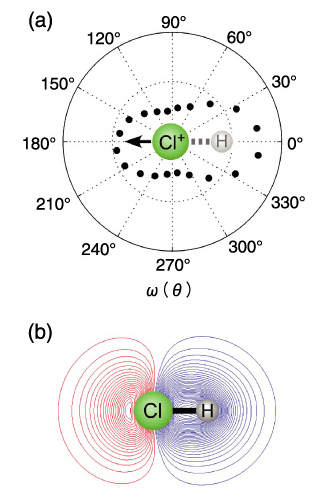
Fig.4-4 (a) Molecular-frame photoelectron angular distribution (MFPAD) obtained for Cl+ ion production. (b) HOMO-1 of HCl, calculated by the full valence CASSCF procedure using the TZV basis sets with polarization functions.
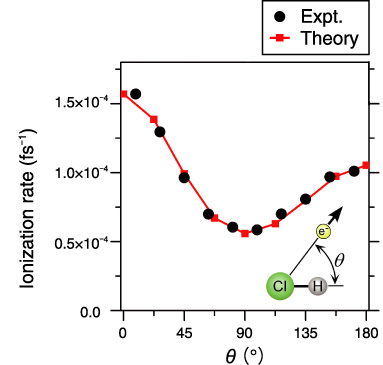
Fig.4-5 Comparison of experimental data and DFT simulation of angular dependence of ionization probability with HCl
In the present work, we observed that electrons can tunnel out of orbitals at a lower energy level than previously realized. Recent advances in ultrafast laser technology enable us to obtain an intense laser field as high as 1015 W/cm2. Since the intensity is comparable in magnitude to the Coulomb field generated by the atomic nucleus, the potential barrier of an atom (or a molecule) is drastically distorted, leading to quantum mechanical tunneling of electrons from the atom or the molecule.
In the case of tunnel ionization from a molecule, scientists have traditionally believed that the electron is ejected only from the highest occupied molecular orbital (HOMO). Ionization from the lower orbitals might be possible, but the probability would be much lower than that for the HOMO, and hence ionization from the lower orbitals has not been observed.
In the present work, we simulated angular dependence of tunnel ionization probability of a hydrogen chloride (HCl) molecule based on the latest computational theory, as shown in Fig.4-5. The simulated result perfectly agrees with the experimentally obtained angular dependence (Fig.4-4(a)), indicating electron ejection from the HOMO-1, the next-highest orbital.
The HOMO of a HCl molecule is a lone-pair 3p orbital of the Cl atom and is not involved in bonding. Thus, the ionization from the HOMO produces a stable HCl+ ion. In contrast, the HOMO-1 is formed by the overlap of the H 1s and Cl 3p orbitals and accounts for the H-Cl bond. Consequently, a HCl+ ion produced by the ionization from the HOMO-1 is unstable and easily fragments. Hence, the breakup of a HCl molecule after ionization is a direct signature for tunneling from the HOMO-1.
By measuring the fragment ion and the tunneled electron in coincidence, we determine the molecular frame photoelectron angular distribution, as shown in Fig.4-4(a), that identifies the HOMO-1 of HCl. An intense, circularly polarized laser pulse singly ionizes and subsequently dissociates an unaligned molecule. The ejected electron drifts perpendicular to the electric field direction at the moment of ionization, while the fragment ion trajectory reflects the orientation of the molecule. The obtained angular distribution (Fig.4-4(a)) is similar to the HOMO-1 (Fig.4-4(b)), and the DFT simulated angular distribution is in nearly perfect agreement with the experimental one, as shown in Fig.4-5.
This research is the first observation of tunnel ionization from lower orbitals than the HOMO. We found that the overall probability for an electron to come from the HOMO-1 is only 0.2%, but if an electron is removed specifically along the molecular axis, away from the H atom, the probability is at least 10%. We expect that tunneling from lower-energy orbitals should be more prevalent in molecules larger than HCl. In such molecules, the energy difference between the HOMO and lower-lying orbitals is generally smaller than that for HCl and would favor lower-energy tunneling.