Table 12-1 Testing conditions
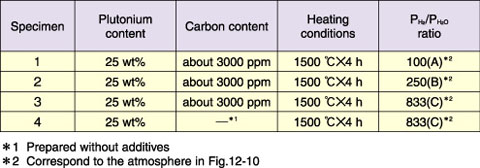
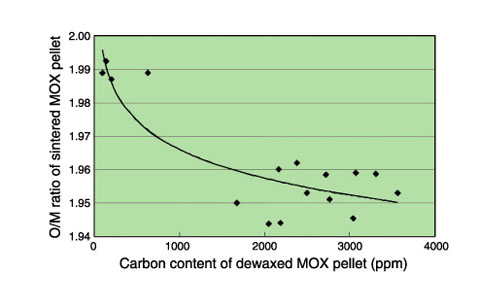
Fig.12-8 Oxygen-to-metal (O/M) ratio vs. carbon content of dewaxed mixed-oxide (MOX) pellets
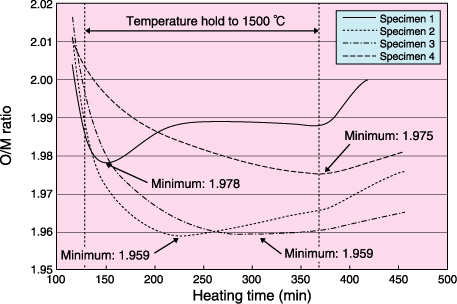
Fig.12-9 Change in O/M ratio during heating
Fig.12-10 Ellingham diagram
We have developed a method based on the “simplified pellet fabrication process” for the production of Mixed-Oxide (MOX) fuels with low oxygen-to-metal (O/M) ratios to control the chemical interaction between the fuels and cladding for a high burnup of 150 GWd/t. However, a substantial amount of time is required to obtain such MOX pellets by sintering and subsequent annealing. In the current pellet fabrication method, which requires the addition of organic additives to MOX powder, about 3000 ppm of carbon remains in the MOX pellets even after pellets are dewaxed at 800 °C. It is empirically known that the O/M ratio of such MOX pellets can be reduced by using the residual carbon after sintering the pellets at 1700 °C (Fig.12-8). Therefore, a new method for preparing MOX pellets having low O/M ratios was investigated; the method involves the use of residual carbon.
The pellets with carbon (specimens 1∼3) and without carbon (specimen 4) were separately heated at 1500 °C in atmospheres having different values of the ratio of the hydrogen partial pressure to the moisture partial pressure (PH2 /PH2O ratio), as shown in Table 12-1. The O/M ratios of each specimen during the heating were estimated from their weight losses. As shown in Fig.12-9, the O/M ratios of the pellets with residual carbon reached the minimum during the temperature hold, whereas the O/M ratios of the pellet without carbon continued to decrease even during the temperature hold. Fig.12-10 shows the oxygen potentials of carbon and the heating atmospheres as a function of temperature. In this figure, the O/M ratios estimated from the oxygen potentials of each atmosphere at 1500 °C are also shown. The oxygen potential of carbon at 1500 °C corresponds to the O/M ratio of 1.939, and is lower than those of the heating atmospheres. Therefore, the O/M ratio of the specimens containing carbon was thought to decrease once toward 1.939 and then increase toward the value corresponding to the oxygen potentials of each heating atmosphere as the residual carbon disappeared in the specimens.
It was found that the influence of the residual carbon was greater than that of the heating atmospheres and that the residual carbon could reduce the O/M ratio in a shorter time. Hence, the addition of carbon to MOX pellets should help in obtaining the desired O/M ratio in large-scale fabrication plants and in overcoming the difficulties associated with the current method such as the inhomogeneous oxygen potentials of sintering atmospheres and the need for a substantial amount of annealing time.