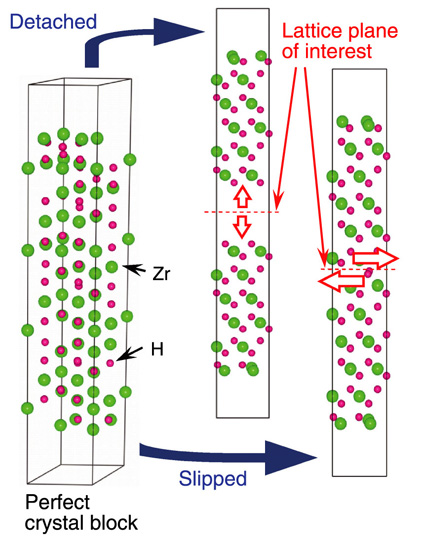
Fig.5-2 Schematic views of first-principles calculations of zirconium hydride
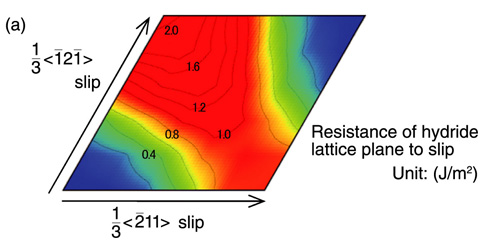
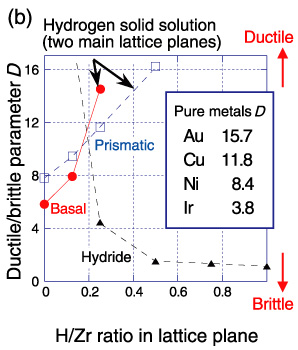
Fig.5-3 Influence of hydrogen concentration on resistance to slip and crack propagation
Hydrogen embrittlement of Zr alloys is an important issue in safety evaluation for nuclear power plants because hydrogen embrittlement results in the mechanical degradation of the fuel cladding. In the reactor operation, the Zr-based cladding is oxidized by waterside corrosion, and it absorbs the generated hydrogen. Only a small part of the absorbed hydrogen is dissolved into the Zr matrix to form a hydrogen solid solution, while the remaining part of the absorbed hydrogen forms zirconium hydride precipitates. Cracks or voids generated inside or around hydride precipitates are known to be the direct cause of observed macroscopic embrittlement. However, it is not microscopically clear how such cracks or voids are generated.
We have performed first-principles calculations to accurately evaluate the microscopic properties of various materials, and thereby to investigate the role of hydrogen in the embrittlement of Zr alloys. The crystal structures of hydride and hydrogen solid solution were modeled (Fig.5-2), and the resistance of their lattice planes to detachment and slip operations were evaluated.
We found that the resistance to detachment in hydride was about 20% lower than that in pure Zr. Further, hydride has a resistance to slip of at least about 1.0 J/m2, irrespective of the slip direction, which is about four-times higher than that of pure Zr and hydrogen solid solution ((Fig.5-3 (a)).
A simple ductile/brittle parameter D considered to be a measure of the resistance to brittle crack propagation was evaluated by using the first-principles calculation results (Fig.5-3 (b)). Generally, D increases with an increase in the resistance to detachment and decreases with an increase in the resistance to slip. The value of D is large for the ductile metal Au and small for brittle metal Ir. The small D value of Zr hydride indicates its crucial brittle nature and implies that crack generation inside hydride precipitates is the origin of the observed macroscopic embrittlement.
Such studies of the elementary processes of hydrogen embrittlement support the ongoing development of safety evaluation methods that can accurately take into account the hydrogen effects. Furthermore, promoting understanding of such influential phenomena on the reactor safety also provides valuable information on how to manage serious situations in nuclear power plants that have not been previously experienced or anticipated.