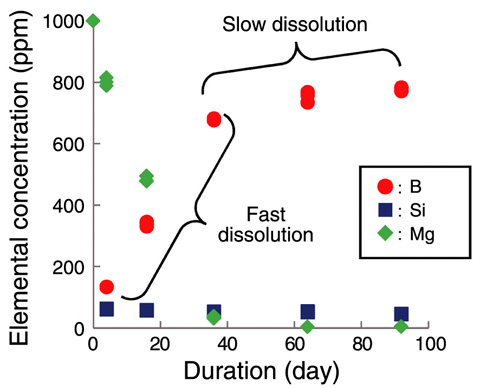
Fig.5-17 Elemental concentrations in the leachate versus time in the leach tests in MgCl2 solution at 90 °C

Fig.5-18 A scanning electron microscope image of the surface of glass leached in MgCl2 solution at 90 °C
High-level radioactive liquid waste generated during the reprocessing of spent nuclear fuel is vitrified into high-level waste (HLW) glass. It has been planned to dispose of HLW glass in geological repositories at a depth of 300 m or deeper by encapsulating HLW glass in an iron overpack and covering it with bentonite clay buffer. Vitrification is one of the methods for the treatment of highly contaminated radioactive waste generated in the aftermath of the accident in Fukushima Daiichi NPS.
It is assumed that after several hundred or several thousand years, the groundwater would permeate the bentonite buffer and corrode the overpack. This may result in radionuclides leaching from HLW glass into the permeating groundwater. For safety assessment of geological disposal, it is important to fully understand the leaching behavior, namely, dissolution behavior, of HLW glass.
Glass dissolves at a high rate when silicon (Si), a glass-network former, in it dissolves. Under stagnant groundwater likely to be present in a deep geological repository, it is presumed that glass would dissolve at a low rate because the concentration of Si in water around glass would reach near saturation.
It is also important to investigate the groundwater components that enhance glass dissolution.
In the present study, static leach tests were conducted on simulated HLW glass in a magnesium chloride (MgCl2) solution to investigate the effect of Mg ions on glass dissolution.
When Mg ions existed in the solution, the glass dissolved at a higher rate (Fig.5-17), and an altered layer including Mg and Si was observed on the surface of the leached glass (Fig.5-18).
The obtained results implied that glass dissolution at a higher rate during the first period of around 40 days was accompanied with the formation of magnesium silicates that consumed glass-network-forming Si. However, after this period, the rate decreased as Mg ions depleted in the solution because Si was not consumed to form magnesium silicates.
In order to understand the dissolution of HLW glass, it is essential to know the elements that are likely to consume Si. Radionuclides would be hardly leached from HLW glass in the long term if such elements are scarce in the groundwater.