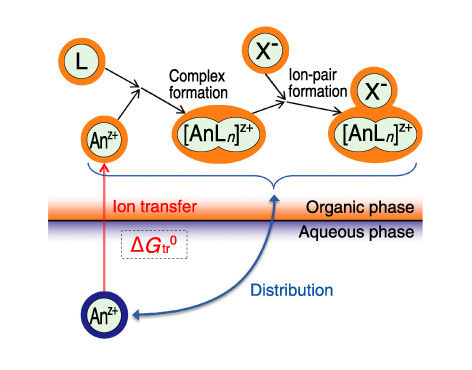
Fig.7-16 Ion transfer at aqueous/organic interface
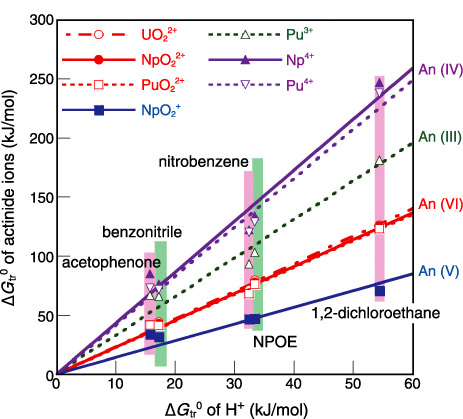
Fig.7-17 Relation between standard Gibbs energies of transfer (ΔGtr0’s) of actinide ions and those of H+
The transferability of an ion at the interface between aqueous and organic phases depends on the chemical potentials of the ion in both phases. Separation of ions by solvent extraction and analysis by using an ion-selective electrode are conducted by utilizing the difference in the ion transferability. Many metal ions require a large energy for their transfer from an aqueous phase to an organic phase (the standard Gibbs energy of transfer at the interface, ΔGtr0) because they are highly hydrophilic and hardly dissolve in an organic solvent. In solvent extraction, ions are extracted into the organic phase by stabilizing them through complex formation and ion-pair formation in the organic phase (Fig.7-16). Accordingly, the distribution of the ions is determined by ΔGtr0 of the ions and counter ions, as well as complex formation and ion-pair formation.
First, we determined ΔGtr0 of the hydrogen ion (H+) by the electrochemical method for use as a reference, in order to estimate ΔGtr0 of actinide ions at the interface precisely. Then, we estimated ΔGtr0 of trivalent- to hexavalent-actinide ions such as uranium, neptunium, and plutonium from the ratio between the concentrations of actinide ion and H+ in both phases for an equilibrium ion distribution.
ΔGtr0 values of actinide ions for five types of organic solvents are plotted against those of H+ in Fig.7-17. It can be observed that the ΔGtr0 values of actinide ions in the same oxidation state are similar to each other irrespective of the type of organic solvent. The ΔGtr0 values of hexavalent actinide ions (AnO22+) are larger than that of the magnesium ion (Mg2+), which is the most hydrophilic among divalent ions whose ΔGtr0 values are known. ΔGtr0 values of actinide ions having a lower charge number are smaller. A comparison of the ratios of ΔGtr0 to the charge number for actinide ions shows that the values of pentavalent neptunium ions (NpO2+) are much larger than those of other actinide ions. Thus, NpO2+ is particularly hydrophilic among actinide ions. It is considered that this high hydrophilicity is one of the reasons why a large distribution ratio of NpO2+ is hardly obtained in any solvent extraction system.
The ΔGtr0 values of actinide ions determined in this study were for organic solvents with relatively large dielectric constants. If more sensitive methods are applied to determine the concentration of actinide ions, the procedure can be extended to organic solvents having low dielectric constants. Accumulated data on ΔGtr0 might be useful for developing guidelines for the selection of a solvent for the extraction of actinide ions and for designing an extraction reagent selective for actinide ions.