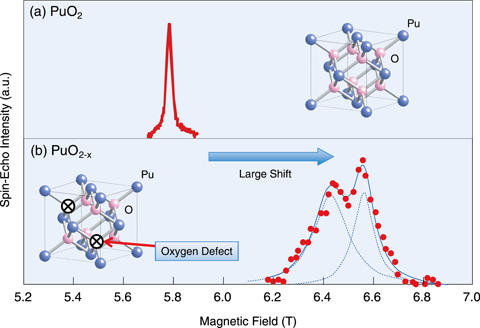
Fig.7-3 239Pu NMR signals observed for the first time in the world
Nuclear magnetic resonance (NMR) can be used to explore electronic states via the interaction between nuclear and electron spins. NMR has been applied to a wide range of fields, such as molecular structure analysis in chemistry and biology, for materials science, and for magnetic resonance imaging (MRI) in medicine. Since an NMR signal was first observed in 1946, it has become possible to detect more than 90 types of nuclear isotopes. Nevertheless, NMR analysis of the actinide nuclei has not been possible, except for 235U. Among the actinide elements, Pu has attracted much attention owing to its noble and mysterious character. Moreover, Pu is important as an atomic-energy fuel, but the electronic state of Pu ions has not been well understood. For such reasons, scientists around the world have been searching for the Pu NMR signal over the past 50 years or more, but until recently, it remained elusive.
We therefore initiated a 239Pu NMR signal search through an international collaboration with the U.S. Los Alamos National Laboratory (LANL). After establishing a detailed experimental design, we succeeded in observing the 239Pu NMR signal for the first time. This success stems from the precise control of many experimental parameters and the use of a very pure sample synthesized by chemists at LANL.
Fig.7-3(a) shows the 239Pu NMR spectrum measured for high purity PuO2. From the field dependence of the central NMR frequency, the nuclear magnetic moment, which is the important physical constant, has been implied to be 0.15 μN (μN is the nuclear magneton). Fig.7-3(b) shows the 239Pu NMR spectrum measured in PuO2-x, which has oxygen defects. When the two spectra are compared, it can be seen that the resonance frequencies and the structures are different. This result implies that the 239Pu NMR analysis has high resolution and is very sensitive to the differences in the coordination state of oxygen.
With this success, it is expected that the research regarding the electronic state of various Pu compounds using 239Pu NMR analysis will now progress. In particular, because the stability of the Pu ionic state in oxides can be judged by elucidating the electronic state of Pu, it is expected that 239Pu NMR will be useful for constructing a safer, prolonged storage method for used atomic-energy fuel containing Pu, which is a globally important issue today.