Fig. 10-2 Effect of buried radioactive waste including nitrate salts
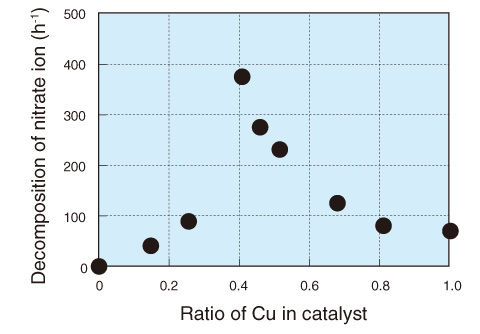
Fig.10-3 Relationship between copper ratio in catalyst and nitrate decomposition rate
When spent nuclear fuel is reprocessed, liquid waste including low-level radioactive materials is generated. The liquid waste includes large amounts of nitrate salts. Because nitrate salts are harmful to human health, the total concentration of nitrate and nitrite in environmental water and drinking water is required to be less than 10 mg/ℓ. When solidified liquid wastes including large amounts of nitrate salts are buried underground, the nitrates in the waste are expected to be eluted into the environment by contact with groundwater (Fig.10-2). Therefore, it is important to remove the nitrate salts before solidification. In industry in general, especially in the field of agriculture, several techniques have been developed for removing nitrate salts at the level of several hundreds of milligrams per liter in agricultural drainage contaminated by fertilization. The nitrate in such waste water is decomposed mainly by using the power of microorganisms in nature. On the other hand, the concentration of nitrate salts in radioactive liquid waste is high, about several hundred thousand milligrams per liter, and a new denitration method is required because microorganisms cannot survive in such harsh conditions. Therefore, we developed a technique for decomposing nitrate by a chemical reaction using a catalyst.
Nitrate ions are reduced to nitrogen using a reductant and a catalyst made of a palladium (Pd) and copper (Cu) alloy. Pd is a precious metal and is expensive, so the performance of the catalyst affects the running cost in practical use. Therefore, a superior catalyst showing such properties as a high nitrate decomposition rate, decomposition of all nitrate ions, little generation of by-products such as nitrogen monoxide and ammonia, and high durability should be developed.
Here we developed a catalyst consisting of micropowders of Pd–Cu alloy supported on the surface of carbon powder and demonstrated its performance. The catalyst was mixed in a sodium nitrate solution including about 300000 mg/ℓ of nitrate ions, and the mixture was stirred and kept at a temperature of 80 °C. We could decompose almost all the nitrate ions by slowly delivering hydradine monohydrate as a reductant dropwise into the mixture. The relationship between the decomposition rate and the Cu ratio in the catalyst was investigated (Fig.10-3), and the maximum decomposition rate was observed when the Cu ratio was around 0.4.
We succeeded in the development of a catalyst that was able to decompose nitrate ions at a high concentration. We will keep developing this technology by enhancing the catalyst’s durability.
<Previous: 10 Development of Decommissioning and Radwaste Treatment Technology | Next: 10-2 >