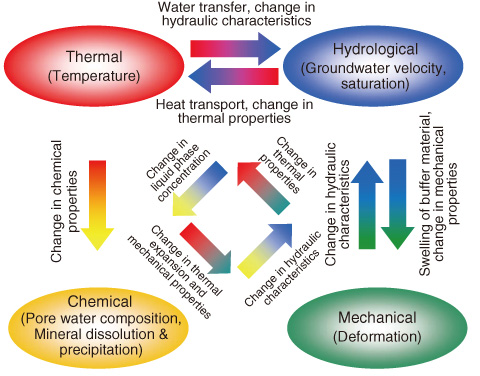
Fig.3-17 Conceptual model of coupled processes in near-field
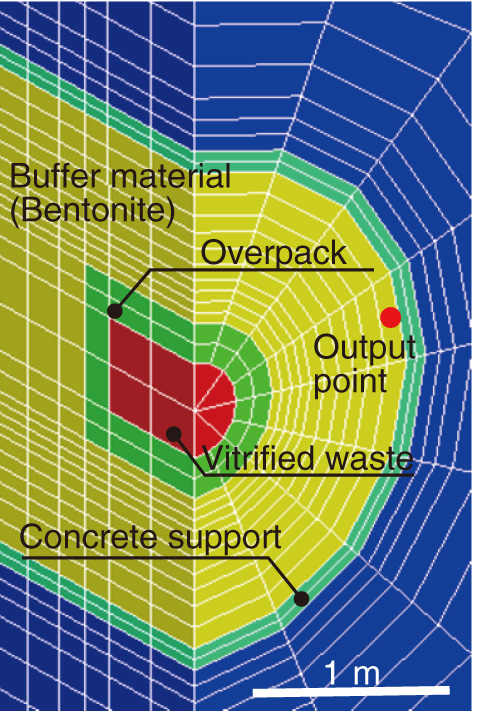
Fig.3-18 Analysis object
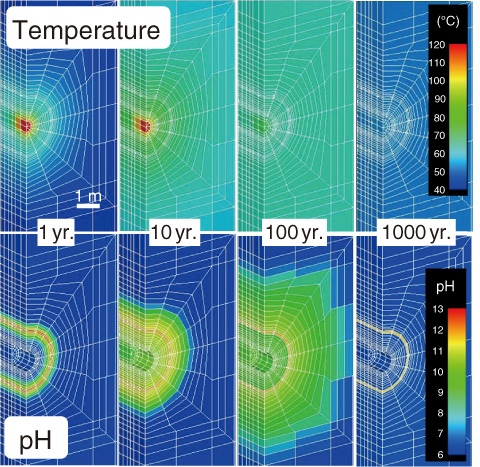
Fig.3-19 Analyzed example: evolution of temperature and pH in the near-field
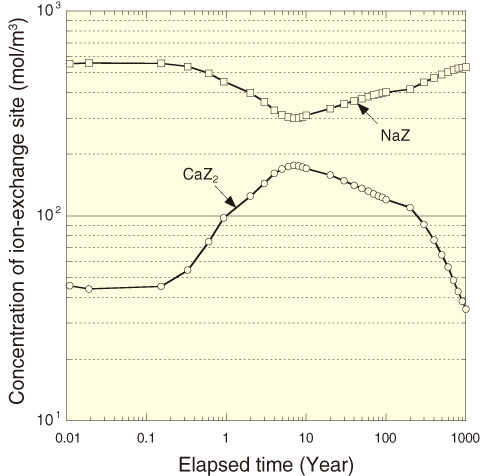
Fig.3-20 Analyzed example: history of concentration of ion-exchange sites in the buffer material
For the safety assessment of the geological disposal of high-level radioactive waste, it is necessary to quantify the coupled thermo-hydro-mechanical-chemical (THMC) processes in the near-field. Because laboratory and in-situ experiments cannot realize real phenomena, numerical models are used as a practical approach to bridging the gap between experiments and real phenomena in long-term near-field evolution, meeting the need for post-closure safety assessment.
This study investigated the geochemical changes arising from the infiltration of groundwater into a bentonite buffer under a thermal regime of radiogenic heating arising from vitrified waste using a computer simulation of a developed THMC model (Figs.3-17, 3-18).
From the analysis results, the temperature in the engineered barrier system (EBS) reached a maximum within 20 years. The temperature of the buffer material on the overpack side was approximately 95 °C. The temperature of the EBS is found to drop to 50 °C after 1000 years. Although the groundwater becomes alkaline under the influence of the concrete support, it returns to the initial pH until after 1000 years (Fig.3-19). In the event of infiltration by saline groundwater, sulfate precipitates as gypsum around the overpack in the buffer material, and the Na-type bentonite is changed to Ca-type by exposure to Ca ions released from the concrete supports (Fig.3-20). These results were consistent with the current scenarios assumed for geological disposal. It will be possible to estimate the extent of the overpack corrosion in the EBS using this developed THMC model.
The present study was sponsored by the Ministry of Economy, Trade and Industry (METI).