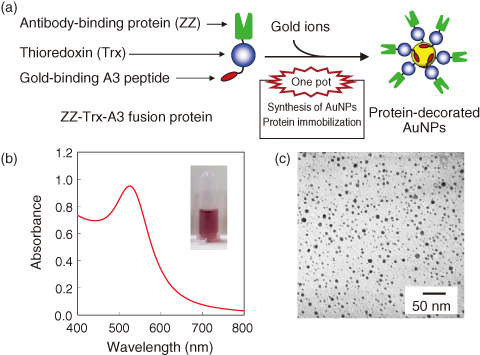
Fig.8-16 Synthesis of AuNPs and immobilization of protein using ZZ-Trx-A3 fusion protein
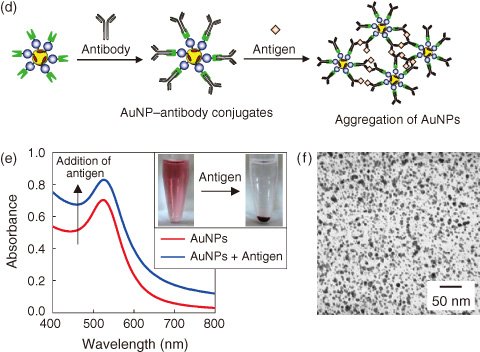
Fig.8-17 Immobilization of antibody on AuNPs and immunoassay
Gold nanoparticles (AuNPs) have received increasing attention in the field of nanotechnology because of their attractive optical properties. AuNP-antibody conjugates are particularly valuable in biomedical applications such as immunoassays for the detection of target antigens and are actually used in a pregnancy test kit. However, the typical fabrication of AuNP-antibody conjugates is time-consuming and requires multiple steps: AuNP synthesis followed by antibody immobilization. In addition, the physical adsorption method, which is commonly employed to immobilize antibodies on the surface of AuNPs, usually yields randomly oriented proteins.
To address this issue, we demonstrate a simple and new strategy for biofabrication of AuNP-antibody conjugates using peptide-functionalized proteins. The strategy is based on a one-pot direct approach that couples peptide-mediated biomineralization and protein immobilization in one simultaneous process. We prepared a tripartite fusion protein (ZZ-Trx-A3) consisting of an antibody-binding protein (ZZ domain), a AuNP-stabilizing protein (thioredoxin) and a gold-binding A3 peptide. By mixing ZZ-Trx-A3 with gold ions, intensely reddish solutions were obtained, and the surface plasmon resonance (SPR) band was observed at 525 nm (Fig.8-16). This indicates the formation of AuNPs from gold ions. Using transmission electron microscopy (TEM), we confirmed that the AuNPs showed spherical morphologies with a narrow size distribution (average diameter 4.4±1.6 nm). The novel method enables facile and rapid fabrication of protein-decorated AuNPs within 20 min and is preferable to conventional methods requiring a few days. In addition, the AuNPs remained suspended in the solution for at least 6 months and in salt solutions adjusted to high concentrations, indicating the high stability of the AuNPs in aqueous media.
As shown in Fig.8-17, protein-decorated AuNPs can be readily derivatized with intact antibodies owing to the specific binding to the ZZ domain on the AuNPs. The addition of target antigens to the AuNP solution yielded a turbid solution owing to AuNP aggregation through the antigen-antibody reaction, which increased the background absorbance. Furthermore, AuNP precipitation was observed after the solution stood for a few hours, and this precipitation was clearly visible to the naked eye.
We expect that our approach can be expanded to produce a variety of biomolecule-decorated AuNPs and can be used in versatile applications in addition to biomedical applications.