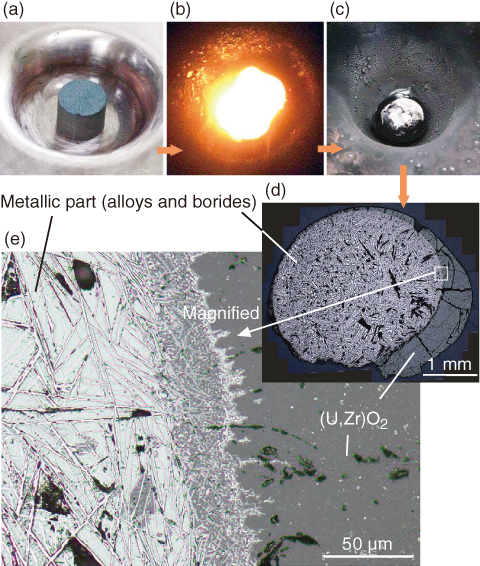
Fig.1-34 The appearance and cross-sectional images of a solidified melt specimens

Fig.1-35 Comparison of the microhardnesses of phases in the solidified melt specimens
For the decommissioning of the accident at the TEPCO’s Fukushima Daiichi NPS (1F), the removal of fuel debris and its appropriate management are imperative missions. Therefore, we have been studying the characteristics of fuel debris using simulated specimens.
The fuel debris formed in the reactor cores mainly comprises materials from the fuel (UO2 and Zr) and control blade (B4C and stainless steel). However, the chemical form of boron in the fuel debris has not been well identified.
To simulate the solidified fuel debris in the reactor cores, some pelletized mixtures of B4C, stainless steel, Zr, and (U,Zr)O2 (a mixed oxide of UO2 and ZrO2) were arc-melted under an Ar atmosphere, as shown in photos (a)-(c) of Fig.1-34. The chemical forms in the solidified specimens were analyzed along the cross section (photos (d) and (e)). The specimens likely consist of (U,Zr)O2 ceramic part and metallic part. The latter is found to include the precipitation of borides expressed as ZrB2 and (Fe,Cr,Ni)2B in the alloy matrix of Fe-Cr-Ni and (Fe,Cr,Ni)2(Zr,U) intermetallic.
To examine the influence of oxygen in the atmosphere, the solidified specimens were annealed at 1500 ℃ in an Ar-0.1%O2 mixed gas flow for 10 h. Consequently, the Zr and U in the alloy, and the Zr in ZrB2 were oxidized to form a Zr-rich (Zr,U)O2 layer on the surface. The boron isolated from ZrB2 alternatively formed (Fe,Cr,Ni)2B inside the oxide layer.
As a basic mechanical property of fuel debris, the microhardnesses of the phases in the specimen were measured by a Vickers tester (Fig.1-35). It is found that the borides, especially ZrB2, are extremely hard compounds in all the phases. The dense precipitation of borides in the fuel debris may potentially be a barrier against machining tools for removal operations.
The present study was sponsored by the Agency for Natural Resources and Energy, Ministry of Economy, Trade and Industry of Japan (METI).