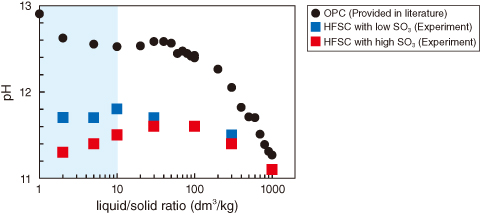
Fig.8-27 Experimental pH value in dissolution experiments
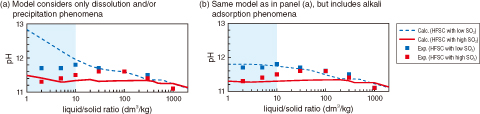
Fig.8-28 Comparison between experimental and calculated pH
Cementitious materials are expected to be used to construct an underground repository for the geological disposal of radioactive wastes. Degradation of ordinary Portland cement (OPC) by the ingress of groundwater can generate a high-pH plume (≥ 12.5), which would probably affect the performance of the bentonite buffer used to contain the radioactive wastes and the transport properties of the host rock. Therefore, a new type of high-volume fly ash silica fume cement (HFSC) is being developed that will lead to a lower plume than for OPC.
The long-term alteration characteristics of HFSC need to be predicted to assist in demonstrating the safety of a geological disposal system. The prediction should be made based on an understanding of the mechanisms responsible for alteration phenomena. In this study, dissolution experiments were used with geochemical equilibrium calculations to make a preliminary assessment of the pH-lowering mechanism in HFSC and to predict the long-term alteration characteristics.
These calculations, based on the understanding that pH decreases only when Ca(OH)2 is absent and the molar Ca/Si ratio in the calcium silicate hydrate (C–S–H) gel is lowered, did not satisfactorily reproduce the experimental results. Dissolution experiments were performed with HFSC containing different amount of SO3, with the focus being on SO42− as a counter ion and on the response of the alkali elements (i.e., Na and K). HFSC containing either high or low SO3 wt% was immersed in ion-exchanged water at various liquid-to-solid ratios, and the resulting pH and liquid composition were measured. At lower liquid-to-solid ratios (≤ 10), the pH was higher for HFSC with low SO3 than for HFSC with high SO3 (Fig.8-27). However, the pH calculated with models that only consider dissolution and/or precipitation phenomena differs significantly from the experimental values for HFSC with low SO3 (Fig.8-28(a)) and so could not be explained by the SO3 content alone.
An improved chemical equilibrium model was developed by including Na and K adsorption on the C–S–H gel along with the dissolution and/or precipitation phenomena. The pH calculated with the improved model agrees well with the experimental results (Fig.8-28(b)). The model thus developed is expected to help improve our understanding of the mechanisms responsible for lowering pH in HFSC and can be used to predict possible scenarios of pH evolution in a geological disposal system.
Part of this study was sponsored by the Ministry of Economy, Trade and Industry of Japan (METI) through the program “Advanced Assessment Technology Development for Impact of Cement on Geological Disposal System for Long-lived Radioactive Waste” in FY 2011 and 2012.