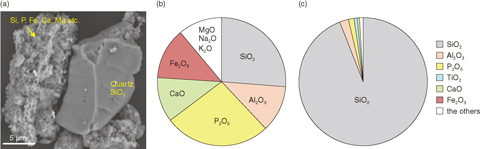
Fig.1-22 SEM image of SSA (a), elemental composition of SSA on oxide basis (b), and elemental composition of residue after SSA was pulverized and treated with a hydrochloric acid aqueous solution (c)
After the accident at the TEPCO’s Fukushima Daiichi NPS (1F), a huge amount of sewage sludge ashes (SSAs) contaminated with radioactive cesium (Cs) (hereafter Cs) was produced at sewage treatment plants in east Japan. Most of them have been stored at those sewage treatment plants. The SSAs containing Cs above 8000 Bq/kg (radioactivity criterion) have been strictly stored as designated waste. Because this was the first case of radioactive contamination of SSA in the world, no scientific knowledge was available to develop a strategy for treatment and disposal of contaminated SSAs.
Si and P were the most abundant elements contained in SSAs, followed by Al, Ca, and Fe (Fig.1-22(a)). SSAs consisted of two main types of particles (Fig.1-22(b)). The first type was natural quartz and feldspar; the second type was a dense aggregate of numerous fine particles thought to be formed during incineration of sewage sludge. These aggregates mainly contained iron oxides, phosphate minerals, and silicon-bearing minerals supposed to be silicon dioxides (hereafter silicon dioxide). We collected SSAs containing Cs of 1F origin above the radioactivity criterion and investigated the relationships among elements such as Cs and iron dissolved from SSAs in acid solution to elucidate the chemical states of Cs, i.e., the solid phase retaining Cs. We found that a significant percentage of Cs was contained as water-soluble salt; approximately 10% was in silicates (quartz, feldspar, and silicon dioxide); and the remainder was in iron oxides. A portion of the iron oxides was not easily dissolved in acid solution, and the Cs concentration in the acid-treatment residue containing iron was not lowered to below the radioactivity criterion because such iron oxide was present inside densely sintered aggregates of numerous nanoparticles with a silicate-framework. Hydrochloric acid solution did not reach the depths of these aggregates. Accordingly, we pulverized the SSAs down to several-hundred nanometers and then dissolved them in hydrochloric acid. Consequently, we succeeded in dissolving almost all of the iron oxides in the SSAs (Fig.1-22(c)). The Cs concentration in the acid-treatment residue of pulverized SSAs was lowered to below the radioactivity criterion. Long-term leaching tests confirmed that the Cs contained in the acid-treatment residue was not leacheable to pure water and seawater at the raised temperature.
We expect that these findings will contribute to the development of a strategy for appropriate treatment of the designated waste containing radionuclides released from 1F and reducing its volume.