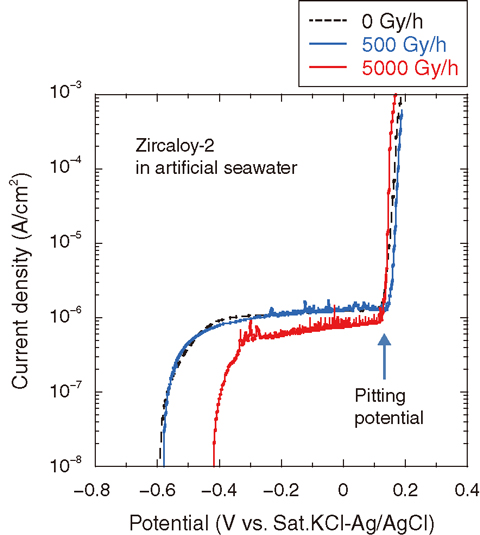
Fig.1-23 Polarization curves of Zircaloy-2 immersed in artificial seawater at ambient temperature
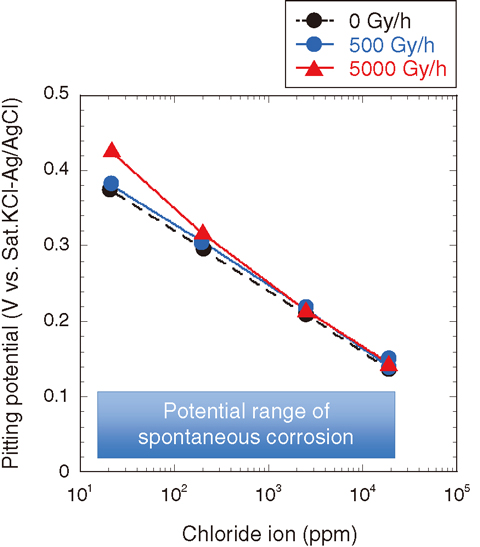
Fig.1-24 Pitting and spontaneous potentials of Zircaloy-2 as functions of the chloride concentration of diluted artificial seawater
At the TEPCO’s Fukushima Daiichi NPS (1F), the cooling function of the spent fuel pools (SFPs) failed with the loss of the electrical power supply, so seawater was injected into the SFPs of Units 2-4 as an emergency measure for avoiding fuel exposure to air. Corrosion troubles are not a concern under circulation of purified water in SFP, but they are under circulation of water contaminated by seawater. Chloride ions, which originate from seawater. can lead to pitting corrosion of fuel cladding tubes (Zircaloy-2) and radioactive substances are leaked from the pitting position. To avoid a leakage risk, TEPCO has been removing seawater components from contaminated water, and fuel rods are cooled by water containing less than 10 ppm of chloride ions.
There are many corrosion survey data for Zircaloy-2 in a clean water environment at high-temperature, but very few data are available in water with a high radioactivity and chloride ion-concentration at ambient temperature.The corrosion data of Zircaloy-2 in the latter environment is needed to evaluate the integrity of Zircaloy-2 cladding tubes in SFPs after accidents. Therefore, we have investigated the electrochemical corrosion behavior of Zircaloy-2 in diluted artificial seawater under γ-ray irradiation at ambient temperature. Pitting and spontaneous potentials of Zircaloy-2 were measured as the electrochemical corrosion data.
Fig.1-23 shows polarization curves of Zircaloy-2 immersed in artificial seawater at ambient temperature. The potential at which the apparent rise of current density occurs is called the pitting potential. There is no difference in the pitting potentials under the tested dose rate conditions (0, 500, 5000 Gy/h). This result suggests that pitting potential is not affected by γ-ray irradiation. Fig.1-24 shows the relationship between the pitting and the spontaneous potentials of Zircaloy-2 and the chloride ion-concentration of diluted artificial seawater. It is known that pitting corrosion can occur if pitting potential is less than spontaneous potentials. Pitting potentials are higher than spontaneous potentials in all cases, indicating that pitting corrosion of Zircaloy-2 hardly occurs in the tested solution.
At present, the chloride ion-concentration of circulation water is maintained at less than 10 ppm in SFPs and the common pool of 1F. It is believed that Zircaloy-2 is not subject to pitting corrosion in the above mentioned environments.