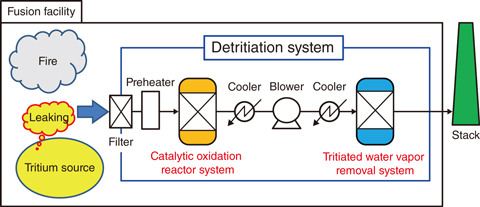
Fig.9-15 A schematic diagram of a detritiation system
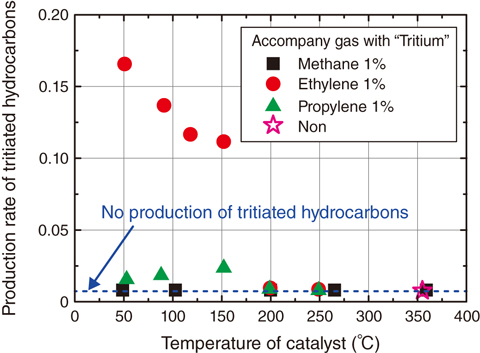
Fig.9-16 Production rates of tritiated hydrocarbons in the case where tritium with hydrocarbons is introduced into the catalytic reactor
A large amount of tritium is used as a fuel in nuclear-fusion facilities. Since tritium is a radioactive isotope of hydrogen, a detritiation system (DS) is installed in fusion facilities to prevent tritium release into the environment. The DS must ensure the detritiation efficiency under all possible extraordinary events. We have investigated the effects of the events upon the detritiation efficiency through experiments and analysis to design a DS that can work efficiently under all extraordinary events.
The DS comprises a catalytic-oxidation-reactor system and a water-vapor-removal system (Fig.9-15). The catalytic-oxidation-reactor system must ensure the tritium-oxidation efficiency in the case where various types of gases with tritium are fed into the system under extraordinary events. Assuming a fire event, hydrocarbons such as methane and ethylene are produced by combustion of flammable organic matter. Issues with the catalytic-reactor system include ensuring the tritium-oxidation efficiency, even when tritiated hydrocarbons are produced, and controlling excessive temperature increase owing to hydrocarbon-combustion reactions, which induce risks of excessive tritium permeation and explosion and so on.
We have clarified that a tritiated-hydrocarbon-production reaction only occurs to a notable extent for ethylene (Fig.9-16). This indicates that the isotopic-exchange reaction between tritium and hydrogen is very slow and that the hydrogenation reaction is the dominant producer of tritiated hydrocarbon. It is found that tritiated methane, which is combusted at a high temperature, is produced only by isotopic-exchange reactions, and also that tritiated hydrocarbons are combusted by the catalytic reactor, whose temperature is sufficient to combust ethylene completely. Therefore, these results suggest that a catalytic reactor at a comparative low temperature of 200 °C ensures both the tritium-oxidation efficiency and the excessive temperature rise by heat of reaction in the catalytic-reactor system.
The Japan domestic agency is in charge of procurement of DS for ITER construction in the south of France within the ITER organization. We are conducting qualification tests of DS and proceeding R&D for safety of a nuclear-fusion facility.