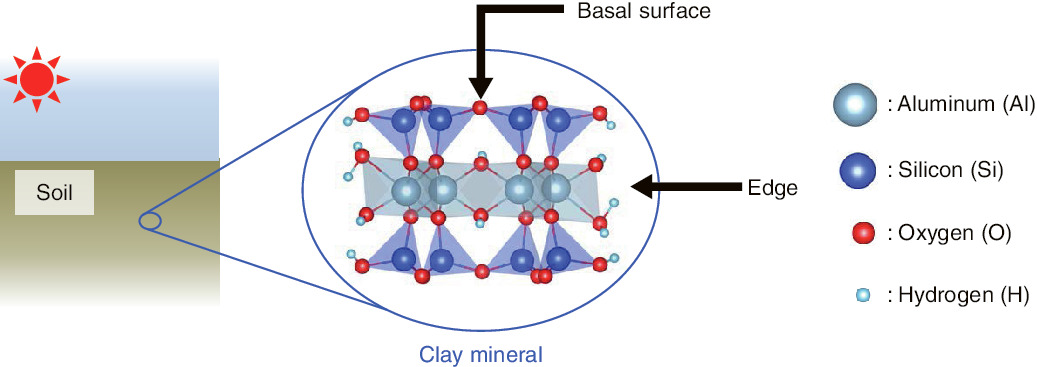
Fig.1-40 Edge structure of clay minerals
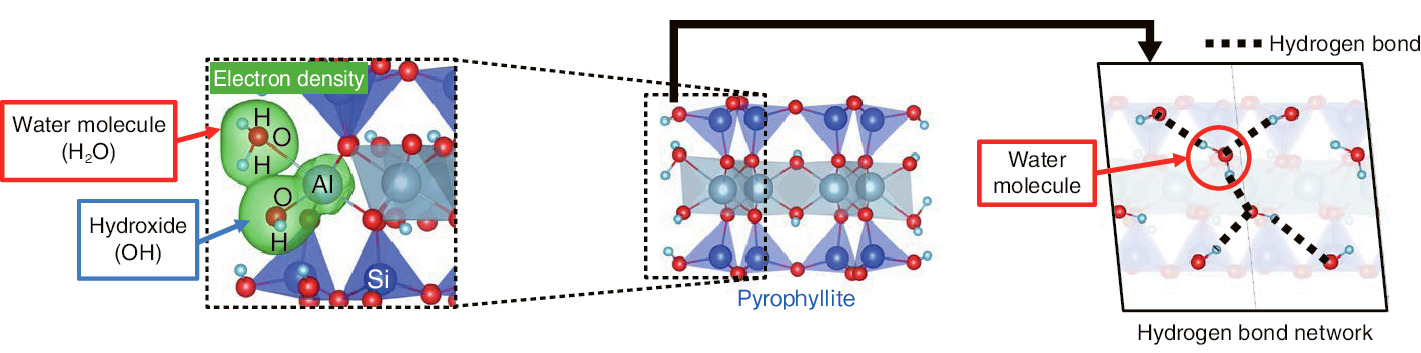
Fig.1-41 Difference in bonds between water molecules and edge Al atoms and between hydroxides and Al atoms
Huge quantities of radionuclides were released into the environment by the accident at the TEPCO’s Fukushima Daiichi NPS (1F). Radiocesium caused evacuation of residents near 1F because it remains on surface soil as a long-lived γ-ray emitter due to its long half-life and strong adsorptive properties. The Japanese Government has performed large-scale decontamination in Fukushima. These activities have been shown to be effective, i.e., the air-dose rate has been reduced. Another issue, however, has arisen: huge amounts of waste are being produced by decontamination. Volume reduction and safe storage of the waste soil is required to reduce the burden to residents of Fukushima. Therefore, extensive research on the development of effective volume-reduction techniques is ongoing.
It is known that radiocesium is mainly adsorbed by clay minerals in soil. If it were possible to remove radiocesium from clay minerals, the waste volume would drastically reduce. However, we lack effective techniques for volume reduction; one reason for this is lack of scientific knowledge concerning cesium (Cs) adsorption to clay minerals. It is believed that radiocesium falls onto the ground surface in raindrops as ions, which are finally adsorbed by clay minerals via organic/inorganic materials in the surface soil. However, the details of the process, particularly at the atomic scale, remain unknown. If the details are clarified, development of waste-soil-volume-reduction techniques should advance.
Long-term study of Cs adsorption to clay minerals has revealed that the edges of such minerals (Fig.1-40) are good sites for adsorption of Cs. However, the detailed atomic-scale structure of the edge has yet to be revealed, because it is difficult to observe its non-crystalline structure even using recent advanced experimental apparatuses. In particular, some numerical simulations have predicted that water molecules exist stably at the edge (Fig.1-40); however, the mechanism behind this phenomenon is not understood. The structures of the edge, including water molecules, are thought to affect the affinity of Cs directly. Therefore, we study chemical bonding between the water molecule and the edge of pyrophyllite, which is the simplest clay mineral, using density functional theory.
We observed that the water molecule bonds to the edge by forming a hydrogen-bond network, although the bond between the hydroxides and aluminum (Al) atoms in the edge is covalent (Fig.1-41). We plan to study the dynamics of the water molecule when Cs comes to the edge. We are continuing our study to develop a technique for waste-volume reduction, which contributes to the reduction of environmental damage.