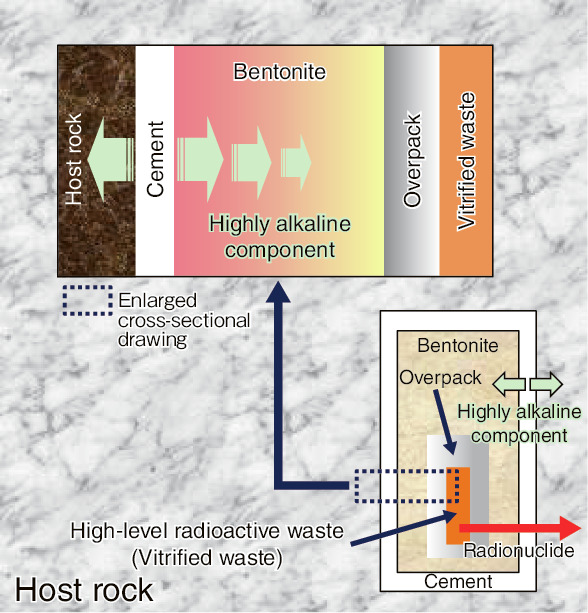
Fig.2-10 Conceptual view of the HLW-disposal system
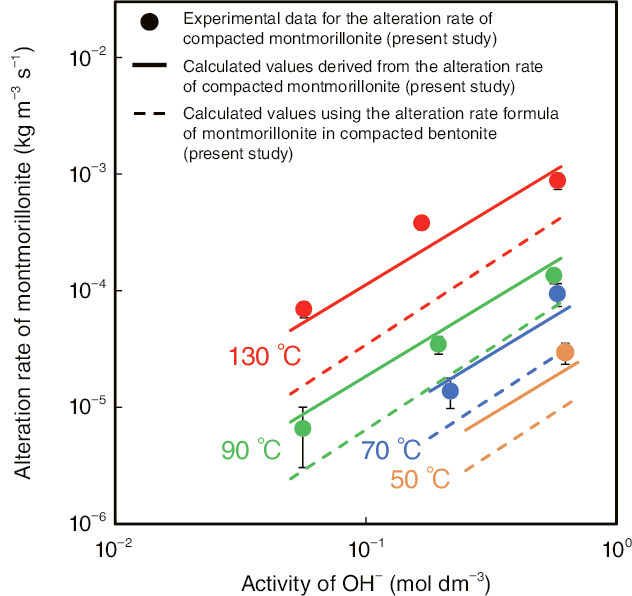
Fig.2-11 Alteration rate of montmorillonite in highly alkaline solutions
In geological disposal system of high-level radioactive waste (HLW), vitrified waste is encapsulated in an iron container called an overpack, surrounded by an engineered bentonite-buffer material (compacted bentonite), and placed in a repository in a stable host rock, as shown in Fig.2-10. Properties of bentonite, high sorptivity and low permeability, retard outward radionuclide migration. As the retardation properties of the bentonite depend upon its montmorillonite content, the alteration of montmorillonite into other minerals induced by the highly alkaline component supplied from the cementitious materials in the system must be quantitatively evaluated in assessments of the long-term retardation properties of the system.
In our previous studies, the alteration rate of montmorillonite in compacted bentonite was experimentally formulated. However, decreases in the concentration of hydroxide ions (OH-) by alteration of accessory minerals and its effect upon the alteration behavior of montmorillonite in compacted bentonite were not clarified.
In the present study, the alteration rate of compacted pure montmorillonite, free of accessory minerals, was experimentally investigated at temperatures of 50 ℃–130 ℃ and a formula was proposed. Then, the alteration-rate formula of montmorillonite in compacted bentonite was compared with that of compacted montmorillonite to clarify the effect of accessory minerals upon montmorillonite alteration.
As shown in Fig.2-11, the alteration rates of compacted montmorillonite (RA) were formulated as a function of the activity of OH-(aOH-) and the absolute temperature (T):
RA=30000·(aOH-)1.3·exp(−55000/RT) [kg m-3 s-1],
where R is the gas constant. The alteration rate of montmorillonite is observed to be approximately 2.5 times higher in pure compacted montmorillonite than in compacted bentonite. This result suggests that the decrease in OH- concentration by reacting with accessory minerals in the bentonite has a great effect upon the alteration rate of montmorillonite and that the OH- concentration in compacted bentonite was overestimated in the previous alteration-rate formula of montmorillonite (RA=3500·(aOH-)1.4·exp(−51000/RT)), i.e., the alteration rate of montmorillonite was underestimated.
The introduction of the alteration-rate formula derived in present study into a geochemical-calculation code under development may make it possible to evaluate the alteration of montmorillonite and accessory minerals simultaneously in compacted bentonite.
The present study was sponsored by the Secretariat of the Nuclear Regulation Authority (NRA) (formerly the Nuclear and Industrial Safety Agency, Ministry of Economy, Trade and Industry (METI), Japan).