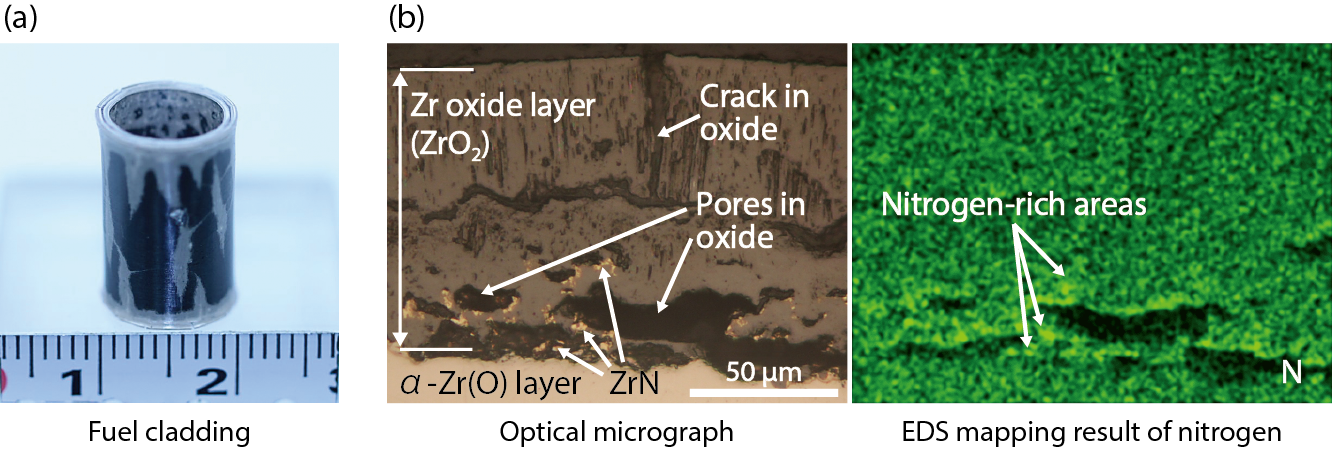
Fig.2-6 (a) Appearance and (b) optical micrograph (left side) and EDS mapping result of nitrogen (right side) of a cross-section of the specimen exposed to steam with a 70 % air fraction at 1273 K for 2400 s
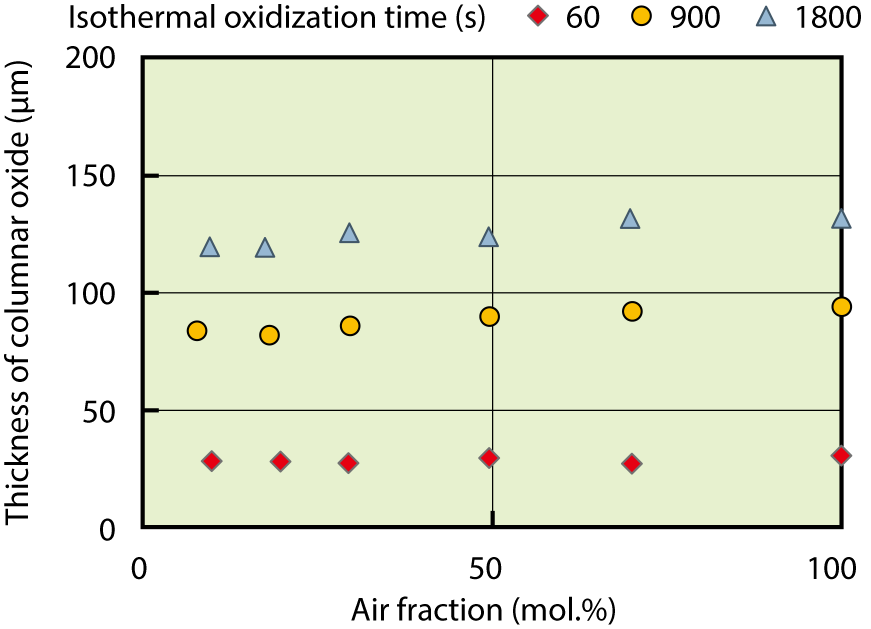
Fig.2-7 Dependence of the thickness of columnar oxide on the air fraction in steam at 1473 K
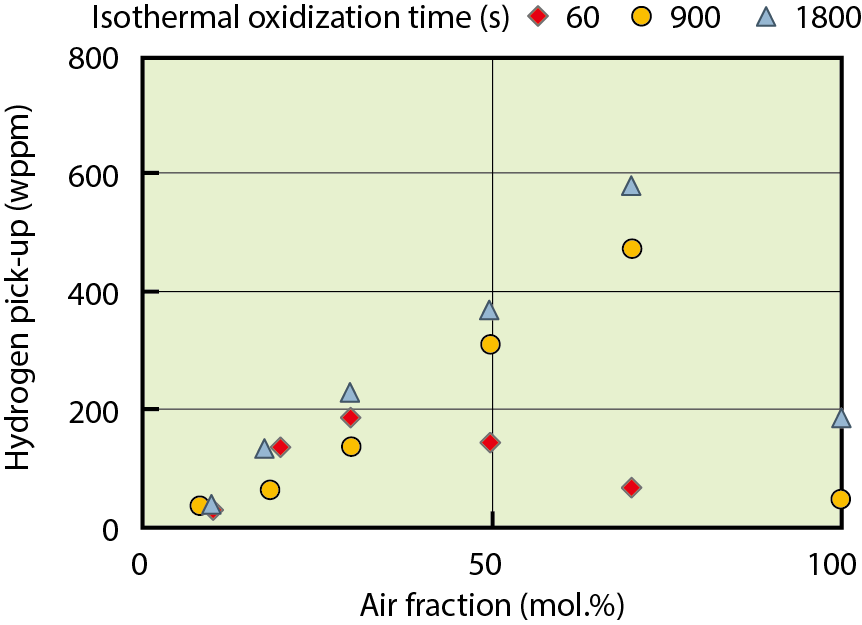
Fig.2-8 Dependence of hydrogen pick-up in the fuel cladding on the air fraction in steam at 1473 K
The generation of heat and hydrogen from nuclear fuel during an accident is strongly affected by the atmosphere surrounding the fuel cladding. Whereas many data have been obtained regarding the behavior and rate of fuel cladding oxidation in high-temperature steam, there has been little knowledge gained in terms of the oxidation behavior under mixed steam-air conditions. In this study, the oxidation behavior of fuel cladding was investigated by heating it under mixed steam-air conditions at temperatures corresponding to loss-of-coolant accidents (LOCA) in the spent fuel pool and reactor core. Air can ingress the spent fuel pool and also the reactor core if emergency core-cooling systems fail during the postulated LOCA, resulting in a severe accident.
Current fuel cladding consists of a zirconium (Zr)-based alloy, and an oxide layer forms on its surface in a high-temperature oxidative atmosphere. An increase in the thickness of this oxide layer, however, may lead to cladding embrittlement and the subsequent fracture of fuel rods in the reactor core. On the other hand, it has been pointed out that nitrogen in the surrounding atmosphere could enhance the oxidation of Zr through nitride formation. In addition, hydrogen is generated during the steam oxidation of Zr at high temperatures. A part of this hydrogen would be absorbed into the metallic part of the oxidized fuel cladding, which would accelerate cladding embrittlement. The oxide layer formation and the acceleration of oxidation and hydrogen absorption into the metallic part by nitrogen would affect the integrity of the fuel rods.
In this study, we investigated the influence of the air fraction in steam on hydrogen absorption and the growth of columnar oxide at 1273 K and 1473 K, subjected to non-irradiated Zry-4 tube cladding. From the results of this study, it was found that the oxidation of the fuel cladding was accelerated by nitride formation near the interface between the oxide layer and metallic part, which is shown in Fig.2-6, and also by the effect of the air fraction in steam on the growth of columnar oxide, which is shown in Fig.2-7.
The amount of hydrogen absorbed into the fuel cladding during high-temperature oxidation is shown in Fig.2-8. It was found that hydrogen absorption was accelerated by an increase of the air fraction in steam. This is because the porosity of the oxide layer increased owing to nitride formation, and the hydrogen that was generated by the oxidation reaction could easily reach the metallic part. On the other hand, the amount of hydrogen absorption decreased in the higher air fraction region owing to the lower partial pressure of steam in the atmosphere, which generated a smaller amount of hydrogen.
We clarified the oxidation mechanism of fuel cladding in an atmosphere with a steam-air mixture at high temperatures.