
Fig.2-16 Analytical flow for individual uranium particles
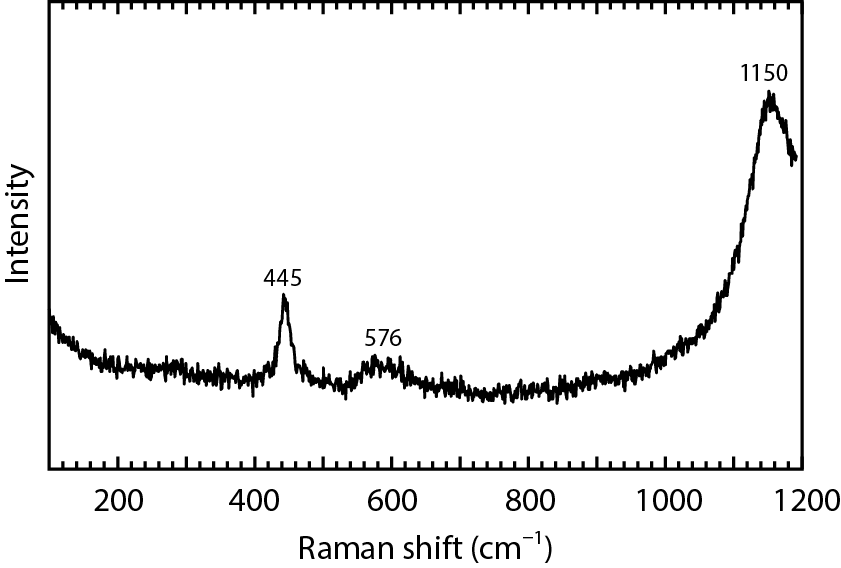
Fig.2-17 Raman spectrum of a uranium dioxide particle
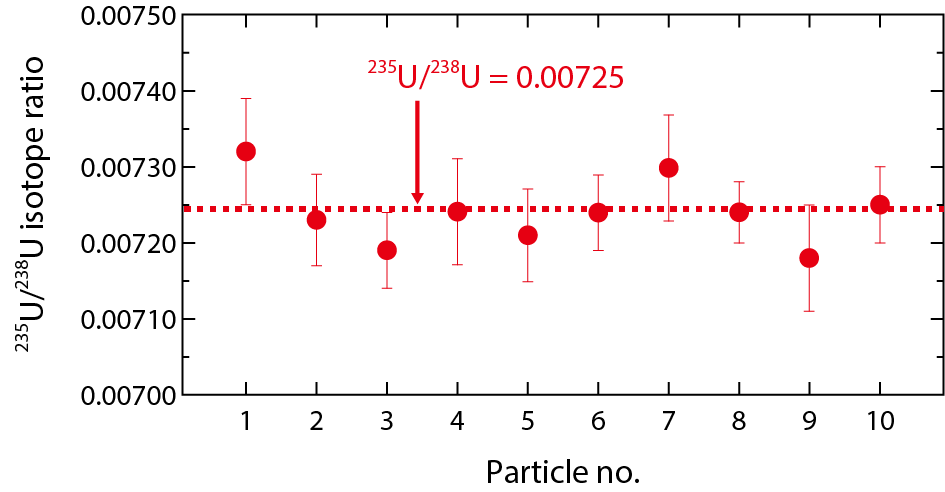
Fig.2-18 235U/238U isotope ratios measured for individual uranium particles
Naturally occurring uranium consists of 234U, 235U, and 238U isotopes. The uranium isotope 235U is enriched to extract nuclear power effectively because it is the only fissile isotope of natural uranium. The abundance of 235U is 0.72 % for natural uranium, 2 %–5 % for nuclear fuels for nuclear power plants, and over 90 % for the raw material for nuclear weapons. We routinely measure uranium isotope ratios in environmental samples, which are collected by International Atomic Energy Agency (IAEA) inspectors at nuclear facilities all over the world, to detect undeclared nuclear activities related to the production of nuclear weapons.
The nuclear fuel cycle is the progression of nuclear fuel through a series of processes. Various uranium compounds are used in the refining, conversion, enrichment, and fabrication processes, including triuranium octoxide in the refining process and uranium dioxide (UO2) in the fabrication process. Investigation of the chemical states and isotope ratios of uranium particles may provide information on the origin of materials. However, there are few reports on the analytical techniques for obtaining both the chemical states and isotope ratios of individual uranium particles.
In this study, we developed a novel analytical procedure using micro-Raman spectroscopy (MRS) and secondary ion mass spectrometry (SIMS), as shown in Fig.2-16. Uranium particles on a substrate were identified by scanning electron microscopy and then transferred onto another substrate using a micro-manipulator. The chemical forms of uranium particles were analyzed by MRS, and the isotope ratios of the particles were determined by SIMS.
Fig.2-17 shows the Raman spectrum of a UO2 particle in a natural uranium material. Characteristic Raman bands were observed at 445 cm-1, 576 cm-1, and 1150 cm-1, which are the values assigned to the UO2 structure. The uranium isotope ratios of individual UO2 particles were determined by SIMS after the MRS measurement. Fig.2-18 shows the isotope ratios of ten individual UO2 particles. The 235U/238U isotope ratios were highly consistent with that of natural uranium. We confirmed that the chemical states and isotope ratios of individual uranium particles could be analyzed by this proposed method.
The present study was sponsored by the Secretariat of the Nuclear Regulation Authority (NRA), Japan.