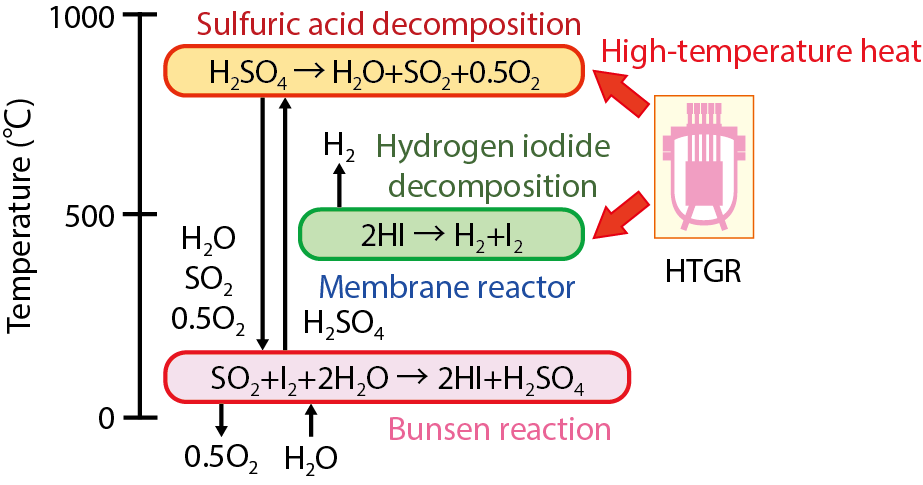
Fig.6-11 Schematic of the IS process
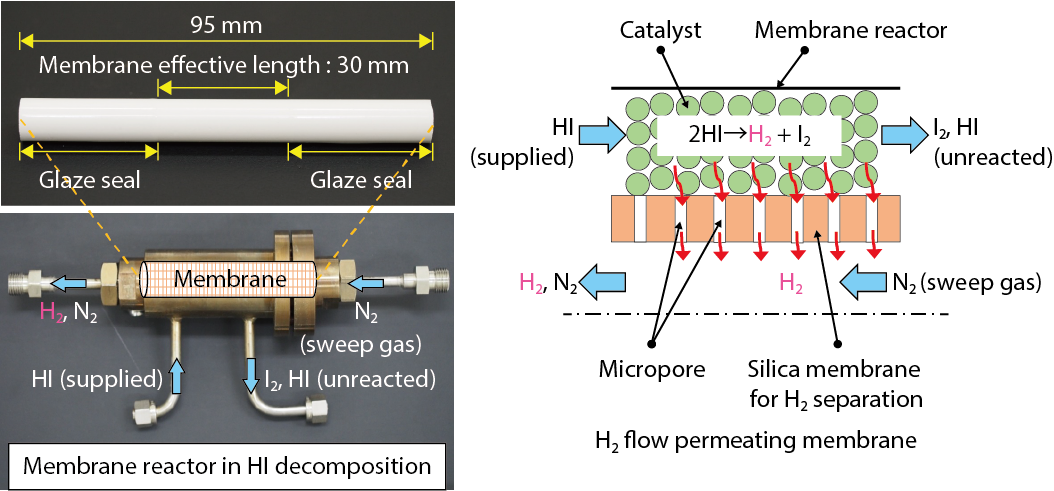
Fig.6-12 Schematic of the membrane reactor equipped with a hydrogen-separation silica membrane in HI decomposition
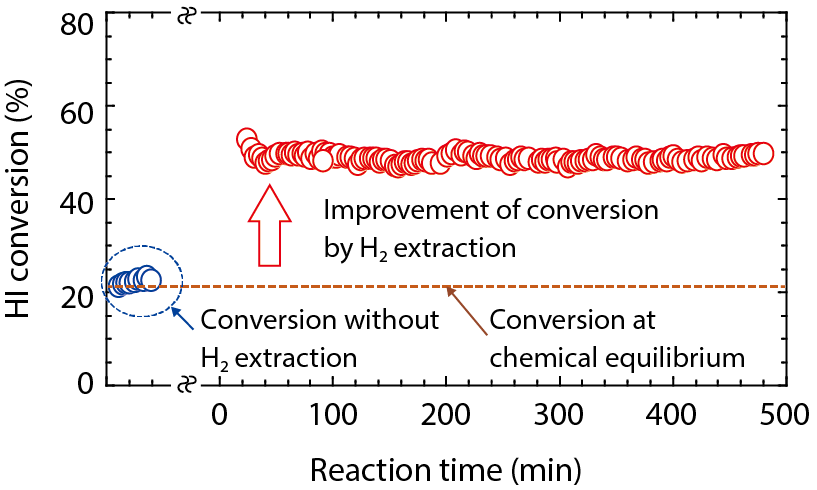
Fig.6-13 Enhancement of HI decomposition conversion by the membrane reactor
With the aim of contributing to a low-carbon society, we are researching and developing a thermochemical IS process for hydrogen generation, which uses the heat generated by a High Temperature Gas-cooled Reactor (HTGR). This thermochemical IS process uses water as its raw material, and iodine (I) and sulfur (S) in its reaction processes (Fig.6-11). As the entire process requires only water and nuclear heat and releases only hydrogen and oxygen, it is expected as a future CO2-free hydrogen production technology.
Since the IS process converts thermal energy into chemical energy of hydrogen, an improvement of the process efficiency is one of the important tasks. The efficiency-determining step in this process is the HI decomposition, owing to its low conversion at chemical equilibrium (approximately 20 % at 400 ℃). This low-decomposition conversion increases the amount of recycled materials, e.g., iodine and HI, thereby increasing the thermal burden and decreasing the thermal efficiency of the total process.
The HI conversion can be improved by a membrane reactor that extracts hydrogen from the reaction field. Removing the hydrogen from the membrane reactor improves the reaction efficiency. To this end, we have been developing elemental technologies of the membrane reactor for HI decomposition.
The membrane is the heart of the membrane reactor. The high decomposition temperature (> 400 ℃) and highly corrosive HI environment exclude the use of common hydrogen-separation membranes such as polymers and palladium membranes. A promising candidate is silica membrane.
We have fabricated high performance silica membranes by chemical vapor deposition on porous alumina supports (Fig.6-12). Fig.6-13 shows the results of the membrane HI decomposition test. The HI conversion was increased from 20 % at equilibrium to approximately 50 %. Removing the hydrogen shifted the reaction equilibrium, promoting the HI conversion. Currently, the expected hydrogen production efficiency of the IS process is approximately 40 %. The targeted efficiency is 50 % in future developments. We are working to improve the thermal efficiency by developing large-scale HI membrane reactor.
The present study was supported, in part, by the Council for Science, Technology and Innovation, Cross-ministerial Strategic Innovation Promotion Program (SIP), “Energy Carrier”, funded by the Japan Science and Technology Agency (JST).