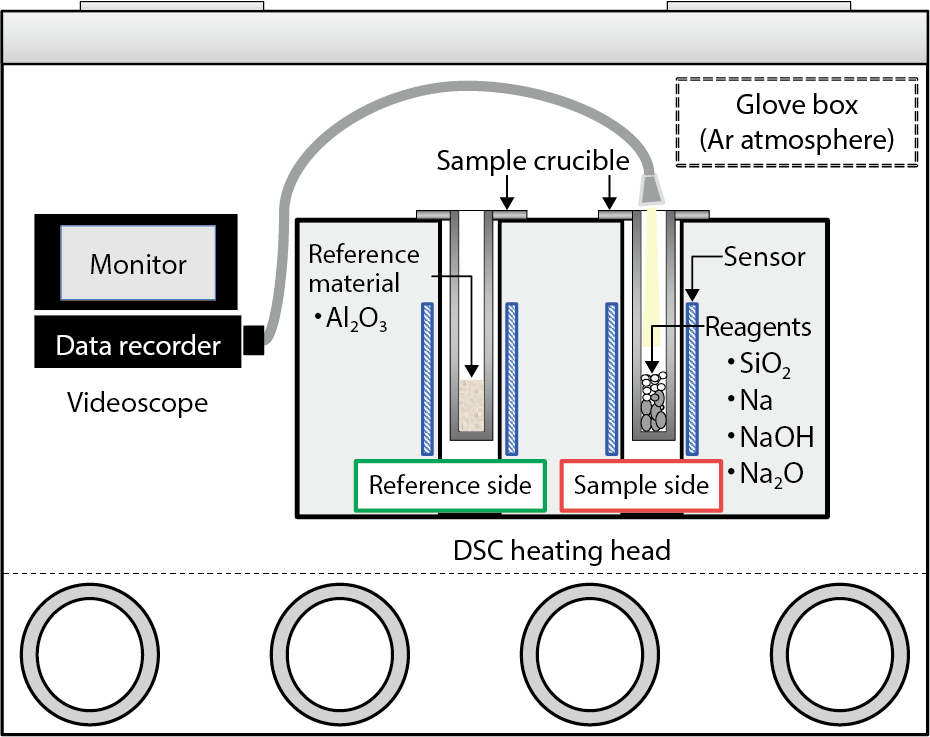
Fig.7-15 Experimental apparatus and situation
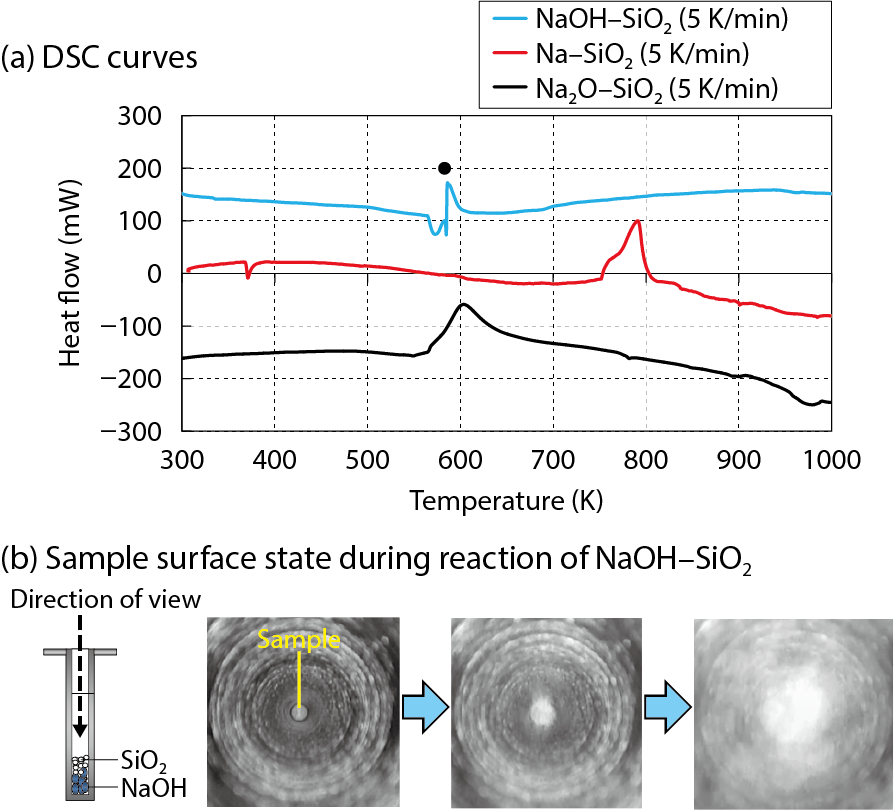
Fig.7-16 Comparison of DSC curves and observations during a reaction
As a countermeasure against sodium leaks in sodium-cooled fast reactors (SFRs), a steel liner is built to protect against the chemical reaction caused by direct contact of liquid sodium with structural concrete. However, if considering severe accidents such as a breach of this steel liner by an intensive sodium leak, a sodium–concrete reaction (SCR) may occur. SCR may result in ablation of structural concrete with exothermic heat and the release of hydrogen gas. Thus, it is important to evaluate the behavior of SCR. Though various experimental studies on SCR have been performed in many countries to obtain findings on this subject, these have not been sufficient to elucidate the mechanism behind SCR due to complex and composite phenomena arising from the thermal and chemical effects. Therefore, a mechanistic evaluation method has been developed by identifying the dominant chemical reaction from the viewpoint of chemical reaction dynamics.
In this study, thermal analysis of silica (SiO2) (one of the major compositions of structural concrete) with sodium (Na) was performed to obtain fundamental information concerning reaction behavior. Based on the obtained data, kinetic evaluation was performed. For thermal analysis, a Differential Scanning Calorimetry (DSC) instrument placed in a glove box with an argon (Ar) atmosphere was used because sodium is chemically reactive (Fig.7-15). Sample mixtures of SiO2 with Na, sodium hydroxide (NaOH), or sodium monoxide (Na2O) as byproducts were used and heated at different heating rates of 3–10 K/min in the temperature range of 298–1073 K.
Fig.7-16(a) compares the DSC curves of reactions of Na–SiO2, NaOH–SiO2, and Na2O–SiO2. The DSC curves for reactions of NaOH–SiO2 and Na2O–SiO2 indicate that those onset temperatures are less than 600 K. In the case of a NaOH–SiO2 reaction, an intensive exothermic peak (●) caused by the solid (SiO2)–liquid (NaOH) reaction was observed right after the NaOH melting, indicating intensive reaction-kinetic behavior. Rapid blowout of the sample was observed during intensive exothermic behavior (Fig.7-16(b)). This phenomenon can be explained by the occurrence of the rapid NaOH–SiO2 reaction and accidental blowout of the reacting system caused by the water vapor produced by the cited reaction. On the other hand, the Na–SiO2 reaction occurs at a temperature more than 100 K higher than the NaOH–SiO2 and Na2O–SiO2 reactions, indicating that the kinetic behavior of the Na–SiO2 reaction is slower than that of the NaOH–SiO2 reaction. In cases of reactions of Na–SiO2 and Na2O–SiO2 at different heating rates of 3–10 K/min, the DSC exothermic peaks systematically shift to higher temperatures with increasing heating rate. Based on this kinetic characterization, activation energies for reactions of Na–SiO2 and Na2O–SiO2, as estimated by the kinetic method, were approximately 231 and 106 kJ/mol, respectively. Thus, the Na–SiO2 reaction was slower than the Na2O–SiO2 reaction. Moreover, the NaOH–SiO2 reaction was dominant and significantly faster than other reactions such as Na–SiO2 and Na2O–SiO2.
As a next step, we are implementing thermal analysis of reactions between sodium or sodium compounds with major compositions of structural concrete except SiO2 and developing a mechanistic evaluation method for chemical reaction behavior.
This study was conducted in collaboration with the Hiroshima University.
<Previous: 7-6 | Next: 8 Research and Development on Fuel Reprocessing, Decommissioning, and Radioactive Waste Management>