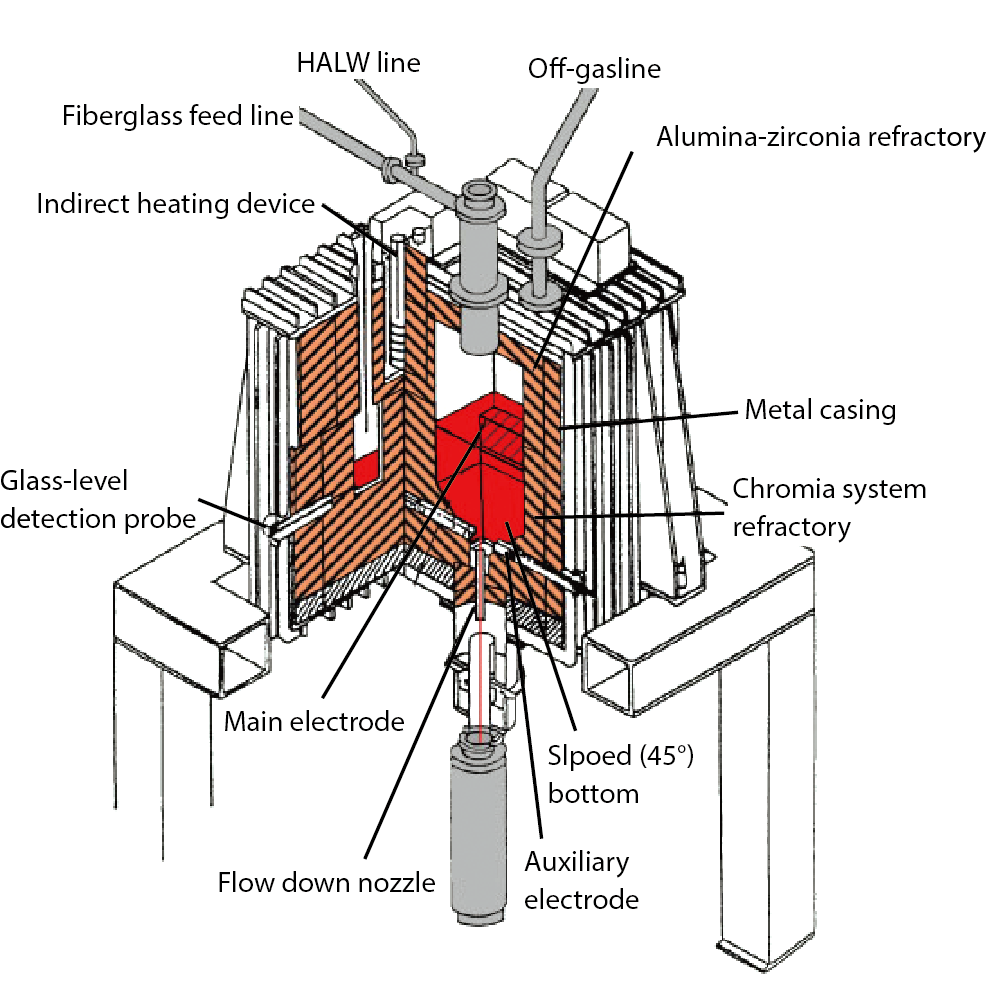
Fig.8-20 Conceptual structure of the melter at the TVF
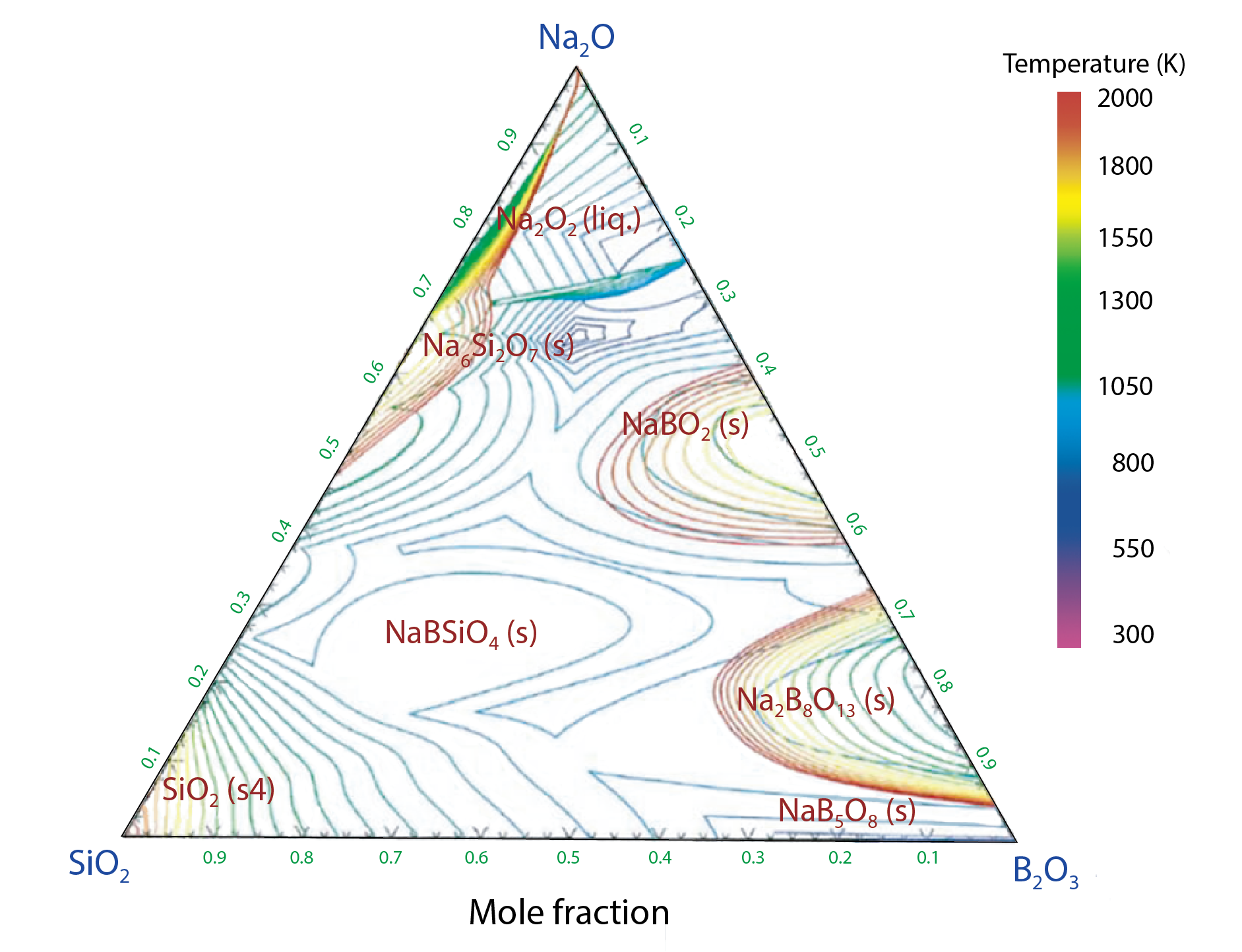
Fig.8-21 Liquidus surface projection for the SiO2-B2O3–Na2O ternary system
The highly active liquid waste generated by the reprocessing of spent fuel is vitrified with using a glass melter at the vitrification facility, as shown in Fig.8-20. The obtained stable vitrified waste is called high-level radioactive waste (HLW) and it is stored until final disposal.
Glass is an amorphous solid that exhibits the glass transition phenomenon with rising temperature (its fluidity increases with a rapid decrease of its rigidity and viscosity). Glass is generally considered to be a fragile material, but it has a high rigidity and a viscosity almost equal to that of crystal when it doesn’t have any defects on its surface. Therefore, in theoretical terms, glass is regarded as one of the toughest materials. Since before the 13th century, borosilicate glass (BSG) has been known to possess a lower thermal expansion, a superior acid resistance, good rigidity, and chemical stability. By the end of the 19th century, BSG was used commercially due to the development of optical glass. As the glass used in HLW production requires the aforementioned properties, BSG is commonly used as the vitrified medium because of the existence of already established technology for its use in HLW production.
Currently, more advance methods are being investigated for the purposes of removal of some fission products (FPs) before vitrification, or to change the ration of glass components, in order to decrease vitrified waste. To minimize radioactivity and the generation of radioactive waste, stable isotopes are commonly used as simulants in vitrification experiments instead of the high-level radioactive FPs, which can lead to the possibility to obtain more data. Before performing the experiment under high temperature, theoretical calculations were used to estimate the thermal behavior of the vitrified waste. As thermodynamic properties are necessary for these theoretical calculations, some missing properties were estimated by computational analysis. The ternary phase diagram shown in Fig.8-21 was constructed as an example.
The search for an enhanced glass medium is carried out gradually with the research and development of vitrification technology. The iron phosphate glasses are an enhanced glass medium which may be able to load various elements and which could prove to be a suitable medium for low-level radioactive waste.
<Previous: 8-8 | Next: 9 Computational Science and E-Systems Research>