Table 1-1 Catalyst specifications
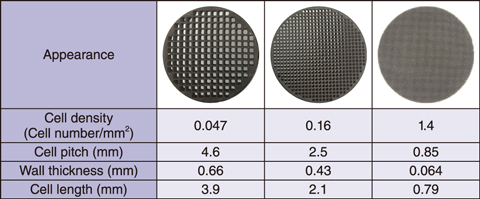
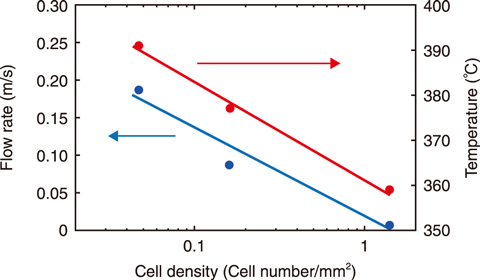
Fig.1-14 Effect of cell density on the catalytic reaction
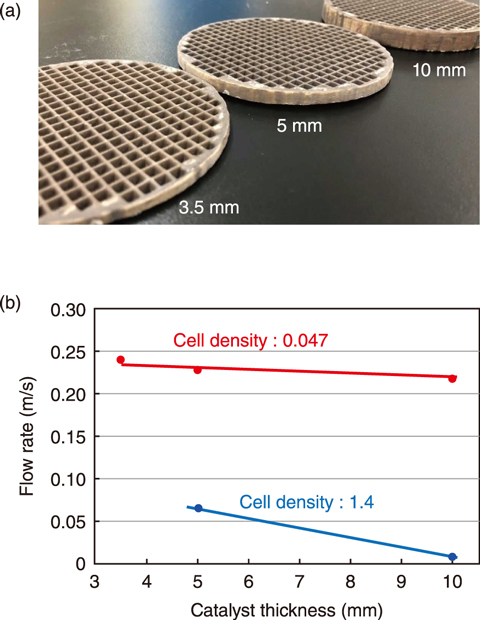
Fig.1-15 Effect of catalyst thickness
Nuclear fuel waste storage containers generate hydrogen gases due to the radiolysis of water molecules. Hydrogen elimination systems using hydrogen recombination catalysts, which do not rely upon an external electric power, have attracted much attention. The catalysts make a non-explosive reaction between atmospheric oxygen and the generated hydrogen, thus reducing the hydrogen concentration without the use of external electric power.
In this work, hydrogen recombination catalysts were developed based on an automotive catalyst and evaluated for their hydrogen elimination capabilities using full-scale equipment. The equipment used for performing the hydrogen recombination experiments was located at the Forschungszentrum Julich in Germany and has a sealed cylindrical shape with an inner diameter of 1400 mm and a height of 3700 mm. After the catalyst was loaded, hydrogen was injected, and the variation in gas temperature, flow rate and hydrogen concentration were observed at several points in the inner space to analyze the hydrogen elimination effect of the catalysts used.
Specifications of the hydrogen recombination catalysts used are detailed in Table 1-1. Precious metal nanoparticles were sprayed on the wall of a monolithic ceramic substrate having many cells. To optimize the catalyst’s shape, several catalysts with different cell densities were prepared. The catalysts were placed in a chimney-shaped tube and installed in the center of the test equipment. The reaction began after hydrogen injection. Because the reaction between hydrogen and oxygen is exothermic, gas convection proceeds upward through the chimney-shaped tube, reducing the hydrogen concentration in the entire space. The catalytic performance was determined by examining the flow rate, as illustrated in Fig.1-14. As the cell density decreased, the flow rate increased, i.e., the catalytic performance improved as the cell density of the catalysts decreased. Several samples with varying catalyst thickness were prepared (Fig.1-15(a)); their resulting catalytic performance is shown in Fig.1-15(b). The flow rate did not significantly decrease when the catalyst had a cell density of 0.047, even when the catalyst thickness was increased. Because the reaction itself increased as the thickness increased, these results indicate that a catalyst with a cell density of 0.047 can be effective for large amounts of hydrogen.
In collaborations with synchrotron radiation experiments and numerical simulations, studies aimed at developing more effective hydrogen recombination catalysts and at ensuring safety in waste storage have been promoted.
This work was part of the result conducted “R&D on technology for reducing concentration of flammable gases generated in long-term waste storage containers”, and supported by the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan.