
Fig.1-16 A test tube used in the sorption experiment of U and Np
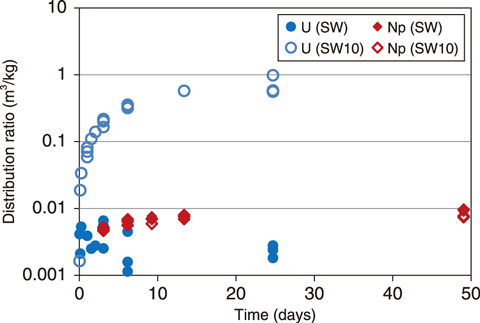
Fig.1-17 Change in distribution ratio of U and Np with time
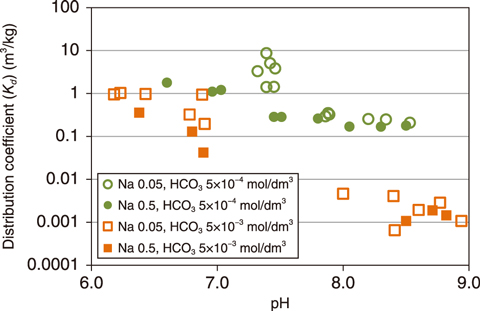
Fig.1-18 Dependency of uranium distribution coefficient on pH
At the TEPCO’s Fukushima Daiichi NPS, contaminated water has been processed by water treatment apparatuses to remove radionuclides. Spent zeolite, used as an absorbing material in these apparatuses, is disposed of as radioactive waste. An estimation of the kinds and amount of the radionuclides present in this spent zeolite is necessary for selecting a suitable disposal concept and safety assessment. However, estimation of the inventory via sampling of zeolite from spent zeolite vessels is difficult because of the high dose rate and vessel structure. Therefore, sorption experiments of U and Np on zeolite (IONSIVTM IE-96, UOP), which is used in the water treatment apparatus SARRY, were conducted to develop an inventory estimation method based on the sorption behavior of radionuclides on zeolite (Fig.1-16).
The resulting distribution ratios using simulated seawater (SW) and simulated seawater diluted ten times by deionized water (SW10) are shown in Fig.1-17. Here, the distribution ratio was calculated by dividing the amount of U or Np sorption on zeolite (i.e., solid phase concentration) by the concentration of U or Np in the solution. A high distribution ratio indicates that a large amount of U or Np was sorbed onto the zeolite. The results indicated that a low distribution ratio of U was obtained in SW, whereas the distribution ratio was significantly increased in SW10. Alternatively, the distribution ratio of Np was low and independent of seawater concentration.
To clarify the dominant factor causing the variation of the distribution ratio of U between SW and SW10, an experiment was carried out as functions of carbonate and Na+ concentration in the experimental solution. The resulting distribution coefficient (Kd) is shown in Fig.1-18 as a function of the pH, where Kd is defined as the distribution ratio at sorption equilibrium. In the pH range from 8 to 9, the Kd of U was low at a higher carbonate concentration, whereas the Kd was independent of Na+ concentration. This indicates that the difference in Kd of U between SW and SW10 is due to the difference of carbonate concentration in the solution. The decreasing Kd with increasing carbonate concentration may be caused by the increase in the concentration of uranium-carbonate complexes, which have a low sorption ability on zeolite. The Kd of Np was found to be independent of Na+ and carbonate concentration. This was consistent with the trend of the distribution ratio of Np observed in the experiment using SW and SW10.
Thus, the carbonate concentration may be a dominant factor in controlling the inventory of radionuclides in spent zeolite.
This work was part of the result conducted “Development of technology for treatment and disposal of accident waste”, and supported by the Agency of Natural Resources and Energy (ANRE), the Ministry of Economy, Trade and Industry (METI), Japan.